|
IMMULITE®
2000 Assays Perform!
Introduction
On March 3, 1998, DPC launched the IMMULITE 2000, a new, high throughput,
continuous random access immunoassay system. The IMMULITE 2000 continues
in the IMMULITE® tradition with a wide menu of high-quality assays based
on the same alkaline phosphatase label and ultrasensitive chemiluminescent
substrate, and employing a centrifugal wash method. The launch menu includes
18 assays; a total of 40 are planned by the end of this year. The results
for a sampling of currently available IMMULITE 2000 assays presented here
demonstrate their reliable performance on this new platform.
Thyroid
Function Testing
Modern thyroid function testing paradigms demand TSH assays which are
classified as third generation, that is, which maintain inter-assay coefficients
of variation of 20% or less at a concentration range of 0.01 to 0.02 µIU/mL.
DPC's IMMULITE Third Generation TSH assay - the first such assay to be
fully automated - has now been replicated on the IMMULITE 2000 system.
The results of a comparison study, depicted in Figure 1, demonstrate the
equivalence of the two methods. In a precision study based on NCCLS EP5
guidelines, a sample with a mean concentration of 0.015 µIU/mL was analyzed
by the IMMULITE 2000 Third Generation TSH procedure over a period of 20
days. Analysis of the resulting data yielded the following statistics.
Intraassay
coefficient of variation = 11.7%
Total coefficient of variation = 15.2%
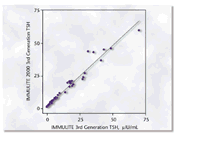 Figure
1. Linear regression analysis of results obtained in a comparison study
using 108 patient samples with TSH values ranging from 0.004 to 70 µIU/mL
yielded the equation IMMULITE 2000 = 0.94 (IMMULITE) + 0.23 µIU/mL with
a correlation coefficient of 0.985. Figure
1. Linear regression analysis of results obtained in a comparison study
using 108 patient samples with TSH values ranging from 0.004 to 70 µIU/mL
yielded the equation IMMULITE 2000 = 0.94 (IMMULITE) + 0.23 µIU/mL with
a correlation coefficient of 0.985.
A
precision-dose profile obtained for multiple samples appears in Figure
2. These data clearly indicate that the IMMULITE 2000 Third Generation
TSH assay yields precision performance which places it well within the
parameters of the third generation TSH definition.
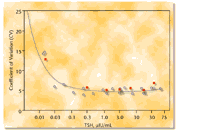 Figure
2. Precision-dose profile for IMMULITE 2000 Third Generation TSH assay,
including results for samples processed in accordance with the NCCLS EP5
guideline. Circles: within-run CVs. Figure
2. Precision-dose profile for IMMULITE 2000 Third Generation TSH assay,
including results for samples processed in accordance with the NCCLS EP5
guideline. Circles: within-run CVs.
Squares:
run-to-run ("total") CVs. Dilutional parallelism constitutes another means
for evaluating assay sensitivity on low-concentration samples. Figure
3 shows observed versus expected concentrations for a low sample that
was serially diluted to the sensitivity of the assay.
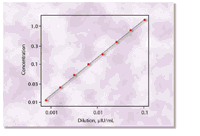 Figure
3. Observed values for a sample containing a low TSH concentration, serially
diluted to the sensitivity of the IMMULITE 2000 Third Generation TSH assay.
The straight line indicates expected values. Figure
3. Observed values for a sample containing a low TSH concentration, serially
diluted to the sensitivity of the IMMULITE 2000 Third Generation TSH assay.
The straight line indicates expected values.
Such
assay sensitivity imparts practicality to the reflexive testing feature
available on the IMMULITE 2000, whereby an option may be chosen to reflex
automatically to another test if results fall outside the reference range.
Samples with high or low TSH values, for example, can automatically undergo
testing for free T4. This feature can enhance the efficiency of algorithms
such as the one employed at Mayo Clinic, in which third generation TSH
serves as the frontline test followed by free T4.
Rounding
out the available thyroid function testing parameters are IMMULITE 2000
Total T4, Thyroid Uptake, Free T4, and Total T3. Future additions to the
thyroid menu will be Free T3, Anti-TPO Ab, Anti-TG Ab, TBG, and Thyroglobulin.
Fertility/Infertility
Testing
One of the hallmarks of DPC is its expansive menu of products designed
to evaluate fertility and infertility parameters. DPC's history in this
testing area began with manual RIAs that later made the technology transfer
to the automated chemiluminescent IMMULITE system. These same high-quality
products now have counterparts on the new IMMULITE 2000 platform. The
available assays include Estradiol, Progesterone, LH, FSH, Total HCG,
Prolactin, and DHEA-SO4, with Total Testosterone and SHBG to follow.
IMMULITE
2000 HCG exemplifies the synchronicity of instrumentation and assay design
features, which combine to provide an amplified assay range of up to 5,000
mIU/mL with no high-dose hook effect even at HCG levels exceeding 1,000,000
mIU/mL. (See Figure 4.) Furthermore, the unique feature of autodilution
allows samples to be run and diluted only if they are above the calibration
range. IMMULITE 2000 will automatically dilute and reassay these samples
if requested, without operator intervention.
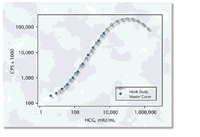 Figure
4. IMMULITE 2000 HCG assay shows no high-dose hook effect even at HCG
concentrations exceeding 1,000,000 mIU/mL. Figure
4. IMMULITE 2000 HCG assay shows no high-dose hook effect even at HCG
concentrations exceeding 1,000,000 mIU/mL.
Tumor
Markers
The worldwide cancer diagnostic market is approximately $1 billion, with
PSA testing alone accounting for nearly a third of this figure. The IMMULITE
system provided the first automated third generation PSA assay, a test
that is now also available on the IMMULITE 2000 platform. An IMMULITE
2000 precision-dose profile appears in Figure 5. These assays have a functional
sensitivity of 0.01 ng/mL (with a detection limit of 0.003 ng/mL), and
provide a tremendous time advantage when monitoring prostate cancer patients.
Other IMMULITE assays for PSA, one with an extended range (up to 150 ng/mL)
and one that detects free PSA,* have also been replicated on the IMMULITE
2000 system. The use of the Free PSA assay in combination with either
of the other two assays for PSA can help elucidate the significance of
elevated PSA levels.
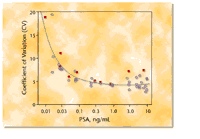 Figure 5. Precision-dose profile for IMMULITE
2000 Third Generation PSA assay, including results for samples processed
in accordance with the NCCLS EP5 guideline. Circles: within-run CVs.
Squares:
run-to-run ("total") CVs.
Figure 5. Precision-dose profile for IMMULITE
2000 Third Generation PSA assay, including results for samples processed
in accordance with the NCCLS EP5 guideline. Circles: within-run CVs.
Squares:
run-to-run ("total") CVs.
Other
tumor marker assays available on the IMMULITE 2000 system include CEA
and, shortly, AFP,* BR-MA (CA15-3),* GI-MA (CA19-9),* and OM-MA (CA125).*
Other
Assays
Another special IMMULITE 2000 feature is the ability to run certain tests
according to barcoded priorities. Whenever the instrument detects that
intact PTH assays, for example, are to be performed, it processes these
tests on all samples on board first (with the exception of STAT samples,
which take precedence), to avoid losses of this temperature-sensitive
analyte. Figure 6 displays the agreement between the IMMULITE 2000 and
IMMULITE assays for intact PTH, as demonstrated in a method comparison
study.
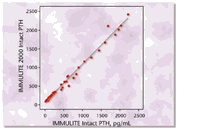 Figure 6. Linear regression analysis of
results obtained in a comparison study using 118 patient serum samples
with intact PTH values ranging from 10 to 2,200 pg/mL yielded the equation
IMMULITE 2000 = 1.05 (IMMULITE) + 1.5 pg/mL with a correlation coefficient
of 0.995.
Figure 6. Linear regression analysis of
results obtained in a comparison study using 118 patient serum samples
with intact PTH values ranging from 10 to 2,200 pg/mL yielded the equation
IMMULITE 2000 = 1.05 (IMMULITE) + 1.5 pg/mL with a correlation coefficient
of 0.995.
IMMULITE
2000 Ferritin is also available as one of the initial launch assays. Additional
assays under development include ACTH, Beta-2 Microglobulin, Cortisol,
C-Peptide, and Total IgE.
Conclusion
Assay results from beta sites demonstrate the successful replication of
IMMULITE assays on the new IMMULITE 2000 system. IMMULITE 2000 offers
an expanding menu and state-of-the-art solutions for operational simplicity,
systems integration, and assay performance.
*Available
outside the US.
|