  |
New
IMMULITE® Reference Range Data for Reproductive Hormones
As
part of its commitment to the reproductive endocrinology testing market,
DPC organizes multisite clinical studies to obtain reference range data
for the relevant hormones on a variety of populations. Two recently completed
studies, conducted in collaboration with investigators in the US and Europe,
have obtained new reference range information on IMMULITE® assays for
five reproductive hormones. The studies provided data on estradiol, FSH,
LH, progesterone and prolactin from carefully selected, normally ovulating
women; and data on cross-sectional, randomly selected pediatric and cord-blood
samples for FSH, LH, progesterone and prolactin. (Earlier studies had
established reference ranges for DPC's IMMULITE Testosterone, SHBG, and
HCG assays.)
Multicenter
Ovulatory Cycle Study
Reference ranges for normally ovulating women were generated using IMMULITE
assays at study sites in Belgium, Germany, The Netherlands, the United
Kingdom and the United States. Blood was drawn for hormone determinations
from over 50 women in apparent good health, ranging in age from 16 to
44 years (median: 31 years).
Many
ovulatory cycle studies have relied on estimates of the last menstrual
period (LMP), and on a sparse sampling of days from that point onwards,
for dating. As a consequence, such studies are plagued with uncertainties
regarding dates and cycle lengths. Even the major landmark--the LH peak--may
be missed entirely for many subjects. This translates into uncertainties
as to how the longitudinal results for each subject should be merged together
for the purpose of deriving group-based reference intervals for phases
or individual days in the ovulatory cycle.
DPC's
multicenter study, by contrast, drew blood samples on a daily basis throughout
one complete cycle for each subject, beginning with the first day of significant
menstrual bleeding. Uncertainties in dating were thus reduced to a matter
of plus or minus a few hours, guaranteeing a database of high integrity
from which to derive group statistics.
The
IMMULITE results showed excellent correlation with the classic ovulatory
patterns. Moreover, LH/FSH ratios--which are used as a tool in the diagnosis
of patients with conditions of androgen overproduction such as hirsutism,
polycystic ovaries/polycystic ovary syndrome and others--were consistent
with those cited in the literature for healthy women.1
The
study also established early follicular phase reference ranges for FSH
and estradiol levels. Hormonal levels at or around the luteal-follicular
transition of the cycle are relevant to evaluating follicular reserve
and predicting the success of assisted reproductive technologies.
Cross-Sectional
Pediatric Study
A study of pediatric reproductive hormone values examined serum samples
from a pediatric hospital and "wellness" clinic in the southwestern United
States using the IMMULITE FSH, LH, Progesterone and Prolactin assays.
The study included 200 samples from children under 10 years of age, as
well as 68 cord-blood samples. Most of the samples had results by each
of the four DPC assays. Statistics based on these results were tabulated
by age groups.
DPC
Publications
Reference range data from the studies are currently posted on DPC's website.
A more detailed technical report (catalog number: ZB157) contains graphical
representations of the ovulatory cycle data, including (for all five assays)
a conventional plot of concentration against cycle day, with day 0 representing
the occasion of each woman's highest LH value. The report also contains
a plot for FSH, estradiol, and progesterone focusing on the luteal-follicular
transition period, with day 1 representing the first day of the new cycle.
Also included are statistics tabulated for relevant segments of the menstrual
cycle.
Another
technical report titled "Hormonal Levels During the Early Follicular Phase
of the Menstrual Cycle" (catalog number: ZB182-A) explores the clinical
utility of FSH, estradiol and progesterone measurements taken on day 2
and day 3 of the cycle.
DPC
is committed to ongoing studies in reproductive endocrinology, and will
continue to gather and publish data as such information becomes available.
To
obtain copies of technical reports, please contact your local DPC Sales
Representative or your National Distributor.
Choosing
a Window into the Ovulatory Cycle
The conventional representation of hormone levels during the ovulatory
cycle relies on knowing, for each subject, the day of the LH surge, an
event that closely approximates the time of ovulation. With this day designated
as day 0, other sampling days for the same subject are given designations
relative to the LH peak, i.e., the number of days before or after ovulation.
(See Figure 1.) Thus, to the right of day 0, positive day numbers (1,
2, etc.) count forward from the first, second, etc. days of the luteal
phase to the end of the cycle. To the left of day 0, negative day numbers
(‚1, ‚2, etc.) count backwards from the last days of the follicular phase
to the beginning of each subject's cycle. Data points corresponding to
the earliest (e.g., follicular phase day 3, in red) and latest days of
one complete cycle are not aligned in this representation because of differences
in phase lengths.
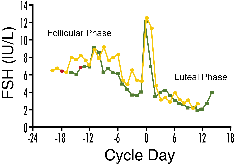 Figure 1. The classic representation of
the ovulatory cycle normalizes hormonal values (in this example, FSH values)
to the day of ovulation. Note that data points corresponding to follicular
phase day 3 (shown in red) for the two subjects pictured are not aligned
in this representation due to differences in their cycle lengths.
Figure 1. The classic representation of
the ovulatory cycle normalizes hormonal values (in this example, FSH values)
to the day of ovulation. Note that data points corresponding to follicular
phase day 3 (shown in red) for the two subjects pictured are not aligned
in this representation due to differences in their cycle lengths.
As
mentioned, it is particularly valuable, from a clinical point of view,
to have reference intervals for both FSH and estradiol during the early
part of the cycle, e.g., for days 2 and 3 of the follicular phase. But
this information cannot be coherently extracted from the conventional
representation: phase and cycle lengths vary from subject to subject,
with the result that days near the beginning and end of the cycles did
not align across subjects.
An
alternative representation which does yield this information is one which
encompasses the last part of the luteal phase of one cycle, and the first
part of the follicular phase of the subsequent cycle. (See Figure 2.)
Such a plot is called a luteal-follicular transition (LFT) plot. Whereas
the conventional representation is centered on the day of the midcycle
LH peak, corresponding to the physiological event of ovulation, the LFT
plot is centered on the divide between one cycle and the next, corresponding
to the inception of significant menstrual bleeding. In the conventional
plot, the follicular phase is on the left, the luteal phase on the right;
in the LFT plot, we see the reverse: the final days of the luteal phase
on the left, the initial days of the follicular phase on the right.
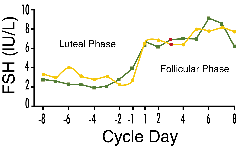 Figure 2. Plot of a portion of the FSH
trajectories for the same two women pictured in Figure 1, normalized to
the luteal-follicular transition. Follicular phase day 3 data points (red)
are aligned in this representation.
Figure 2. Plot of a portion of the FSH
trajectories for the same two women pictured in Figure 1, normalized to
the luteal-follicular transition. Follicular phase day 3 data points (red)
are aligned in this representation.
In
the LFT plot, positive day numbers (1, 2, etc.) count forward toward the
right, beginning from the first day of the follicular phase, while on
the left, negative day numbers (‚1, ‚2, etc.) count backwards from the
last day of the luteal phase. There is, however, no day 0, since there
is no day which separates the end of the luteal phase from the beginning
of the subsequent follicular phase.
Because
of the way the LFT plot is constructed, it is evident that all results
for the clinically important early days of the follicular phase are aligned,
as are the last days of the luteal phase. So, for example, results for
day 3 of the follicular phase, shown in red, are now aligned. As the distance
from the perimenstrual period increases, either backwards towards midcycle
of the first cycle or forwards towards midcycle of the second cycle, the
LFT plot becomes increasingly confused, due to subject-to-subject differences
in phase length. For this reason, it is natural to restrict the LFT plot
to plus or minus 8 days (or less) from the onset of significant bleeding.
Reference
1.
Koskinen P, Penttil”, T-A, Anttila L, Erkkola R, Irjala K. Optimal use
of hormone determinations in the biochemical diagnosis of the polycystic
ovary syndrome. Fert Steril 1996;65:517-22.
|
 |