|
Diabetes
Mellitus
and the Utility of Insulin, C-Peptide and Urinary Albumin
Assays
 Glucose
plays multiple essential roles in life processes. It is a major metabolic
precursor for structural components of DNA and RNA, and serves as the
primary energy source for virtually every living organism. In humans,
glucoregulatory mechanisms normally maintain plasma glucose levels within
a relatively narrow concentration range (70 to 150 mg/dL), despite wide
variations in glucose influx and efflux associated with meals and exercise.
Inadequate glucose levels (hypoglycemia) can cause profound brain dysfunction
and death. Chronic, abnormally elevated levels (hyperglycemia) can lead
to a number of microvascular diseases that can severely damage eyes (retinopathy),
kidneys (nephropathy), nerves, and blood vessels. Glucose
plays multiple essential roles in life processes. It is a major metabolic
precursor for structural components of DNA and RNA, and serves as the
primary energy source for virtually every living organism. In humans,
glucoregulatory mechanisms normally maintain plasma glucose levels within
a relatively narrow concentration range (70 to 150 mg/dL), despite wide
variations in glucose influx and efflux associated with meals and exercise.
Inadequate glucose levels (hypoglycemia) can cause profound brain dysfunction
and death. Chronic, abnormally elevated levels (hyperglycemia) can lead
to a number of microvascular diseases that can severely damage eyes (retinopathy),
kidneys (nephropathy), nerves, and blood vessels.
Defects
in insulin secretion and/or insulin action are the most common failures
in the regulation of plasma glucose levels. Insulin deficiency causes
hyperglycemia, a condition referred to as diabetes mellitus. The word
diabetes means an excessive discharge of urine, and the term mellitus
means honey. Diabetes mellitus refers to metabolic disorders that are
characterized by abnormally elevated levels of sugar in the blood (hyperglycemia)
and urine. Hypoglycemia is most often iatrogenic and is usually associated
with side effects of diabetes treatment regimens.
Diabetes
is a major cause of morbidity and the fourth leading cause of death in
the US. In 1992, diabetics constituted 4.5 percent of the US population
(approximately 11 million people); the disease thus represents a major
public health issue.1,2
Long-term
complications of diabetes mellitus include retinopathy, the leading cause
of adult blindness; and nephropathy, the leading cause of end-stage renal
failure. Diabetes is also a major cause of gangrene, myocardial infarction
and stroke, which result from the associated increased incidences of atherosclerotic,
cardiovascular, peripheral vascular and cerebrovascular diseases.3-6
The
cost of diabetes-associated expenditures to individuals and society is
enormous. In 1992, these expenditures accounted for $105 billion of the
total US healthcare cost, representing approximately one in seven healthcare
dollars spent.7
Classification
of diabetes mellitus
The revised 1997 American Diabetes Association classification scheme8
identifies two major classes of diabetes mellitus: type 1 and type 2.
These designations replace the historic nomenclature--insulin-dependent
diabetes mellitus (IDDM) and non-insulin-dependent diabetes mellitus (NIDDM),
respectively.
Type
1 represents approximately 5 to 10 percent of all diabetes cases. Patients
present with abrupt onset of symptoms that include polyuria, polydipsia,
and rapid weight loss, with peak incidence in early childhood and adolescence.
Destruction of pancreatic islet cells, usually by an autoimmune process,
leads to absolute insulin deficiency. These patients require insulin administration
to sustain life and prevent ketosis.
Type
2 occurs mostly in patients older than 40 years of age. These patients
have minimal symptoms, are not prone to ketosis, and represent approximately
90 percent of all diabetics. The abnormality is associated with impaired
physiological effectiveness of insulin: levels may be normal, decreased
or increased. Control of hyperglycemia may be achieved by weight loss,
and may require treatment regimens involving specialized diets, ingestion
of oral hypoglycemic agents or administration of insulin.
Diagnosis
of diabetes mellitus
The diagnosis of diabetes mellitus depends solely on the demonstration
of hyperglycemia; glucose measurements therefore serve as the front-line
laboratory test. According to the 1997 revised diagnostic criteria,8
one of the following three conditions must be identified, and then confirmed
on a subsequent day:
| A
random plasma glucose > 200 mg/dL (> 11.1 mmol/L) coupled with classic
diabetic symptoms such as polyuria, polydipsia, and unexplained weight
loss. |
| A
fasting plasma glucose > 126 mg/dL ( > 7 mmol/L) after no caloric
intake for at least 8 hours. |
| A
plasma glucose > 200 mg/dL 2 hours after a glucose load (oral glucose
tolerance test; 75 g dissolved in water). |
The
armamentarium of diabetes-related laboratory tests is much broader, however.
It includes tests for insulin, C-peptide, urinary albumin (microalbuminuria),
glycated proteins, associated autoantibodies and genetic markers. These
important laboratory tools are used for screening at-risk individuals,
for characterizing disease status, or for long-term management of diabetic
patients.
The
focus here is on the associated physiology and clinical utility of three
analytes that are generally measured by immunoassay: insulin, C-peptide
and urinary albumin.
Insulin
and C-peptide
Elevated blood glucose levels stimulate pancreatic beta cells to produce
and release insulin into the circulation. Insulin is a polypeptide hormone
that exerts an immediate hypoglycemic effect by promoting cellular uptake
of glucose in muscle and adipose tissue, and by suppressing endogenous
glucose production. Its function is to shift excess extracellular glucose
to intracellular storage sites, mostly in the form of glycogen. Counterregulatory
hormones--glucagon, epinephrin, growth hormone and cortisol--have an opposite
effect. They increase plasma glucose levels by stimulating the breakdown
of glycogen to glucose (glycogenolysis) and the synthesis of glucose (gluconeogenesis).
The
immediate precursor of insulin is proinsulin (MW 9 kDa), a single-chain
polypeptide consisting of 86 amino acids with three disulfide bridges.
Proinsulin is the major storage form of insulin in the Golgi complex of
the pancreatic beta cells. Proteolytic cleavage produces insulin (MW 6
kDa), which consists of 51 amino acids in two chains joined by two disulfide
bridges; and the connecting peptide (C-peptide; MW 3 kDa), a single polypeptide
chain containing 31 amino acids. (See Figure 1.) Equimolar amounts of
insulin and C-peptide are then secreted into the circulation. Circulating
C-peptide concentrations are approximately 5- to 10-fold higher than those
of insulin, however, as a result of the much longer half-life of C-peptide
(approximately 35 minutes, compared to 5 to 10 minutes for insulin).
Human
Proinsulin
Figure 1. Proinsulin undergoes enzymatic cleavage to yield
insulin and C-peptide.
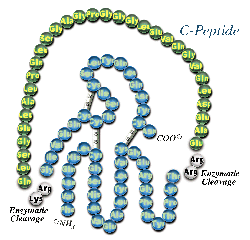
For
an enlarged view, click on the image.
The primary clinical application of insulin and C-peptide measurements
is in the differential diagnosis of fasting hypoglycemia. Elevated levels
suggest an insulinoma. However, elevated insulin results may be due to
factitious hypoglycemia (possibly surreptitious insulin administration),
which must be ruled out before results are accepted as evidence for an
insulinoma. C-peptide measurements are used to resolve the uncertainty.
Levels are low in cases of exogenous insulin administration--commercial
insulin preparations do not contain C-peptide--but are elevated in hyperinsulinemia
resulting from pancreatic secretory activity.
Insulin and C-peptide measurements may be relied on to distinguish between
type 1 and type 2 diabetes by assessing pancreatic reserve.9
Furthermore, insulin determinations have been used for distinguishing
between insulin-requiring type 2 diabetics and type 2 diabetics whose
disease can be controlled with diet and exercise alone. Subjects with
peak plasma insulin levels greater than 60 µIU/mL during a glucose tolerance
test have been reported to be unlikely to develop microvascular complications
and likely to be controlled by diet alone. In contrast, peak insulin levels
below 40 µIU/mL reportedly identify patients who require insulin treatment
and have a higher probability of developing microvascular disease.10
C-peptide determinations have been used in a similar manner. For
example, glucagon-stimulated C-peptide levels greater than 1.8 ng/mL have
been reported to identify type 2 diabetics who could be managed without
insulin treatment. In contrast, levels less than 0.5 ng/mL reportedly
identify type 1 patients who require insulin treatment.11
C-peptide measurements, both basal and glucagon- or glucose-stimulated,
have also been used to reevaluate the necessity for continuing insulin
treatment regimens in patients who may be better managed with diet alone.
Furthermore, the demonstration of glucagon- or glucose-stimulated residual
beta-cell function in type 1 diabetics, as evidenced by a significant
rise in C-peptide values, is considered a better prognostic indicator
than no C-peptide response at all.
Insulin-treated patients present special problems, as insulin assays do
not distinguish between exogenous and endogenous insulin. Furthermore,
such patients are likely to have developed anti-insulin antibodies, which
interfere with insulin immunoassays. Results may be either falsely elevated
or falsely decreased, depending on the characteristics of the insulin
assay. In contrast, C-peptide assays provide a reliable indication of
beta-cell function despite the presence of exogenous insulin and insulin
antibodies.
Urinary
albumin (microalbuminuria)
Diabetes is the most common cause of renal failure in the US. Both type
1 and type 2 diabetics are at high risk for developing diabetic nephropathy
with the associated threats to quality of life and survival. Overt nephropathy
is likely to progress within 5 years to end-stage renal failure requiring
dialysis or renal transplantation.12
Historically, diabetic nephropathy was recognized only when routine urinalysis
revealed protein levels exceeding the detection limits of these assays,
i.e., 200 to 300 mg/L. However, approximately 17 years ago, immunoassays
for measuring urinary albumin became available which allowed for far more
sensitive determinations. These opened up a new window onto the diabetic
kidney, exposing diabetic nephropathy as a continuum that starts long
before urinary protein levels exceed 200 to 300 mg/L.13
The term microalbuminuria was coined to refer to the early phase of diabetic
nephropathy which is characterized by abnormally elevated albumin excretion
rates; albumin levels nevertheless remain below the 200 mg/L threshold.
Microalbuminuria has been defined14 in
terms of abnormally elevated excretion rates, albumin/creatinine ratios,
or albumin concentrations as follows:
| Albumin
excretion rate: |
20-200
µg/min
(30-300 mg/24hr) |
| Albumin
/ Creatinine ratio: |
30-300
mg/g |
| Albumin
concentration, first voided morning urine: |
30-300
mg/L |
Many
studies have confirmed that microalbuminuria is an early predictor of
the renal and cardiovascular complications of diabetes.15
It identifies at-risk patients before major organ damage has taken place,
allowing for early intervention with preventative and cost-saving measures.
For example, intensive blood glucose control has been reported to reduce
the incidence of microalbuminuria and progression to overt renal disease.16
Blood
pressure is an important, modifiable risk factor for the progression of
renal disease. Antihypertensive treatment regimens postpone end-stage
renal failure in patients with overt diabetic nephropathy. Furthermore,
initiation of early antihypertensive treatment lowers urinary albumin
excretion rates and appears to postpone or prevent progression to nephropathy.
Although final conclusions await the completion of long-range studies,
indications are that treatment strategies guided by urinary albumin determinations
can improve patient outcome, save lives, and allow significant reduction
in associated healthcare costs.17 Not
surprisingly, recommendations for routinely assessing microalbuminuria
in diabetic patients at least once a year, if not more often, have gained
wide acceptance.13,14
Summary
Diabetes mellitus comprises a group of common but serious and costly disorders
characterized by chronic hyperglycemia due to defective insulin secretion
and/or action. The two major classifications of the disease are type 1,
which involves pancreatic beta-cell destruction, usually by an autoimmune
process; and type 2, the more common, which is characterized by the impaired
physiological effectiveness of insulin.
Diabetes mellitus is diagnosed by the demonstration of hyperglycemia through
the use of random or fasting plasma glucose determinations, or by an oral
glucose tolerance test. Once diabetes is diagnosed, assays for insulin
and C-peptide can be used to differentiate type 1 from type 2 diabetes
and, among type 2 diabetics, to distinguish those who require insulin
treatment from borderline cases who can be managed with changes in diet
and exercise alone. Insulin determinations are also useful for identifying
patients at risk of developing the complications of diabetes, such as
microvascular disease.
Measurement of C-peptide, a by-product of insulin production, provides
an index of endogenous insulin production and pancreatic beta-cell function.
C-peptide levels, unaffected by exogenous insulin or insulin autoantibodies,
can also be used to distinguish between insulinoma and contrived hypoglycemia,
such as might be caused by surreptitious insulin injection.
Detection of microalbuminuria can identify diabetic patients at risk for
renal and cardiovascular complications before the occurrence of major
organ damage. Lowering albumin excretion rates with antihypertensive therapy
or other measures can either postpone or prevent progression to clinical
nephropathy, thereby saving lives, improving patient outcomes, and reducing
costs.
Blood tests for insulin and C-peptide as well as urinary albumin tests
are commonly performed by immunoassays that provide valuable information
to assist the physician in the identification, characterization and management
of the diabetic patient.
DPC offers assays for the measurement of insulin, C-peptide and urinary
albumin on the automated IMMULITE® platform.
Assay characteristics are summarized below.
|
-
|
Sensitivity
|
Calibration
Range
|
Normal
Reference Range
|
Cross-
reactivity
|
|
IMMULITE
Insulin (enzyme immunometric assay)
|
2
µIU/mL
|
up
to 400 µIU/mL
|
6
to 27 µIU/mL (central 95% range)
|
None
with C-peptide, proinsulin
|
|
IMMULITE
C-Peptide (competitive enzyme immunoassay)
|
0.3
ng/mL (99 pmol/L)
|
0.5
to 14 ng/mL (166 to 4,634 pmol/L)
|
0.9
to 4 ng/mL (298 to 1,324 pmol/L) (central 95% range)
|
None
with insulin, 13% with proinsulin
|
|
IMMULITE
Albumin (competitive enzyme immunoassay)
|
0.5
µg/mL
|
2.5
to 60 µg/mL
|
ND
to 18 µg/min (absolute range, 95% < 13 µg/min)
|
None
with AFP
|
References
1. Prevalence and incidence of diabetes mellitus: United
States, 1980-1987. MMWR Mortality and Morbidity Weekly Report 1990;39:809-12.
2. Harris MI. Impaired glucose tolerance in the U.S. population. Diabetes
Care 1989;12:464-74.
3. Nathan DM. Long-term complications of diabetes mellitus. N Engl J Med
1993:328:1676-85.
4.
Davis MD. Diabetic retinopathy: a clinical overview. Diabetes Metab Rev
1988;4:291-322.
5. Geiss LS, Herman WH, Goldschmid MG, DeStefano F, Eberhardt MS, Ford
ES, et al. Surveillance of diabetes mellitus: United States, 1980-1989.
MMWR Mortality and Morbidity Weekly Report 1993;42:1-20.
6. National Diabetes Advisory Board. Diabetes in the 1980¹s: challenges
for the future. US Department of Health and Human Services, Public Health
Service. NIH Publication No. 82-2143. Washington, DC: Government Printing
Office, 1982.
7.
Rubin RJ, et al. Health care expenditures for people with diabetes mellitus,
1992. J Clin Endocrinol Metab 1994;78:809 A-F.
8. American Diabetes Association. Position statement: Report of the expert
committee on the diagnosis and classification of diabetes mellitus. Diabetes
Care 1997;20:1183-201.
9. Katzeff HL, Savage PJ, Barclay-White B, Nagulesparan M, Bennett PH.
C-peptide measurement in the differentiation of type 1 (insulin-dependent)
and type 2 (non-insulin-dependent) diabetes mellitus. Diabetologia 1985;28:264-8.
10.
Turkington RW, Weindling HD. Insulin secretion in the diagnosis of adult-onset
diabetes mellitus. JAMA 1978;240: 833-6.
11.
Hoekstra JBL, van Rijn HJM, Thijssen JHH, Erkelens DW. C-peptide reactivity
as a measure of insulin dependency in obese diabetic patients treated
with insulin. Diabetes Care 1982;5:585-91.
12.
Hans-Henrik P, et al. Diabetic nephropathy. In: Brenner MB, ed. The kidney.
5th ed. Philadelphia: WB Saunders, 1996:1864.
13.
Watts NB. Albuminuria and diabetic nephropathy: an evolving story. Clin
Chem 1991;37:2027-8.
14. Mogensen CE, et al. Prevention of diabetic renal disease with special
reference to microalbuminuria. Lancet 1995;346:1080-84.
15. Messent JWC, Elliott TG, Hill RD, Jarrett RJ, Keen H, Viberti G. Prognostic
significance of microalbuminuria in insulin-dependent diabetes mellitus:
a twenty-three year follow-up study. Kidney Int 1992;41:836-9.
16. The Diabetes Control and Complications Trial Research Group. The effect
of intensive treatment of diabetes on the development and progression
of long-term complications in insulin-dependent diabetes mellitus. N Eng
J Med 1993;329: 977-86.
17. Borch-Johnsen K, et al. Is screening and intervention for microalbuminuria
worthwhile in patients with insulin dependent diabetes? BMJ 1993;306:1722-5.
Additional
Reference
Sacks DB. Carbohydrates. In: Tietz textbook of clinical chemistry. 3rd ed. Burtis
CA, Ashwood ER, editors. Philadelphia: W.B. Saunders, 1999: 750-808.

|