  |
Prostate
Specific Antigen -
New Developments Beyond the Prostate
Editor's
note: Dr. Diamandis provides here a summary of his slide presentation
at DPC's luncheon symposium during the AACC/ASCLS National Meeting in
Chicago August of 1998.
It
is claimed in the literature that prostate-specific antigen (PSA) is produced
exclusively by the epithelial cells of the prostate gland. Nonprostatic
PSA has been found, however, in hyperplastic and cancerous breast tissue;
all breast secretions (milk, nipple aspirate fluid, and breast cyst fluid);
female serum; lung, ovarian, and salivary gland tumors; and amniotic fluid.
Recent clinical research with PSA has helped to define the role of PSA
in the management of various nonprostatic cancers, mainly breast tumors.
Recent molecular research has revealed abnormalities in the PSA gene in
various cultured tumor cells, and their effect on PSA gene expression.
These molecular findings may contribute to future clinical research.
Clinical
Applications
Cancer has been found more frequently in the female breast and the male
prostate than in any other human tissue.1
In our research, an ultrasensitive time-resolved immunofluorometric assay
(TRIFA) for total immunoreactive PSA has been used to analyze serum samples
from postradical prostatectomy and breast cancer patients.2
The immunoreactivity and sensitivity of DPC's IMMULITE® Third Generation
PSA assay are similar to those of our assay. Very low PSA levels in serum
samples obtained from postradical prostatectomy patients have been detected
by these two assays but not by conventional methods. The increased sensitivity
of these assays affords earlier detection of PSA following prostatectomy.
Further study will be required to determine whether earlier detection
can be expected to produce improved clinical outcome. In breast tumors,
an association has been found between the presence of PSA and steroid
hormone receptors for progesterone and estrogen. We have also developed
an immunoluminometric version of our TRIFA. Free PSA in serum may be a
more specific marker of breast tumors than total immunoreactive serum
PSA. PSA in serum from normal women is found mainly bound to alpha1-antichymotrypsin3
and, in its free form, in serum from women with both benign and malignant
breast tumors.4 Total immunoreactive
serum PSA is not correlated with patient diagnosis or tumor levels, and
does not decline after surgery.5 The
level of expression of PSA is related, however, to a poor response to
tamoxifen therapy in recurrent breast cancer.6
In subgroups of patients with low, intermediate, and high PSA levels,
response rates to tamoxifen were 71%, 51%, and 39%, respectively (P <
0.01).
The
presence of PSA in tumor cytosol may be a favorable prognostic indicator
for women with breast cancer.7 Women
with PSA-positive tumors showed improved disease-free and overall survival
over women with PSA-negative tumors. (See Figure 1.) In addition, PSA
is associated with estrogen and progesterone receptor positivity, younger
patient age, earlier disease stage, smaller tumor size, diploid tumors,
and tumors with low S-phase function.
Figure
1. Prognostic value of PSA in the overall and disease-free survival
of breast cancer patients.
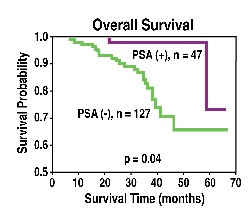
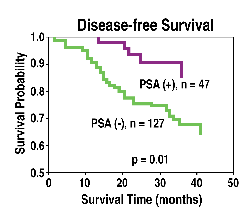
PSA
in breast secretions was found to vary with the condition of the patient
and specimen type.8 PSA at concentrations
up to 300 ng/mL was present in the milk of lactating women. PSA is also
present in milk from prolactinoma patients. In breast discharge fluid,
levels up to 4,000 ng/mL have been detected. In nipple aspirate fluid,
PSA levels as high as 2,000 ‚ 4,000 ng/mL have been found, as compared
to less than 4 ng/mL in male serum. PSA levels in nipple aspirate were
inversely correlated with breast cancer risk.
The
usefulness of prenatal PSA determinations has been explored. Elevated
serum PSA has been found in pregnant women.9
PSA has also been found in amniotic fluid, where its significance is not
yet understood.
Molecular
Studies
Various techniques for studying PSA at the tissue, protein, and genetic
levels have been used in our research. PSA has been localized immunohistochemically
in normal breast tissue, hyperplastic epithelial tissue, papillomas, apocrine
metaplasia, and fibroadenomas. HPLC methods have been used for separating
PSA subfractions and, in addition to Western blotting, for verifying molecular
weight. Direct methods have been developed that measure only free PSA.
At
the genetic level, reverse-transcription polymerase chain reaction (RT-PCR)
has been used to study the expression of PSA in both tumor samples10-12
and tissue culture model systems.13,14
PSA gene sequencing has been used to determine genetic aberrations that
may lead to altered expression of the PSA protein. The genetic transcript
of PSA (PSA mRNA) from breast tumors shared identical sequence homology
with PSA mRNA from the prostate. Levels of PSA protein produced by these
tumor cells have been found to range from undetectable to very high. This
varied level of protein production led to the study of the genetic basis
of the expression of PSA protein. Genetic alterations were found only
in regions of the PSA gene that do not code for the PSA protein but enhance
its expression.10,11 PSA gene expression
in cultured breast carcinoma cells was also found to be upregulated by
androgens and progestins.
Further
study of the physiology of PSA in the breast is required to understand
its significance in this tissue. Since PSA is an enzyme, it must have
a natural substrate in breast tissue. The connection between PSA expression
and the pathophysiology of breast cancer remains to be established.
References
1.
Lopez-Otin C, Diamandis EP. Breast and prostate cancer: an analysis of
common epidemiological, genetic and biochemical features. Endocr Rev 1998;19:365-96.
2. Ferguson RA, Yu H, Kalyvas M, Zammit S, Diamandis EP. Ultrasensitive
detection of prostate-specific antigen by a time-resolved immunofluorometric
assay and the IMMULITE immunochemiluminescent third-generation assay:
potential applications in prostate and breast cancers. Clin Chem 1996;42:675-84.
3.
Melegos DN, Diamandis EP. Diagnostic value of molecular forms of prostate-specific
antigen for female breast cancer. Clin Biochem 1996;29:193-200.
4. Borchert GH, Melegos DN, Tomlinson G, Giai M, Roagna R, Ponzone R,
Sgro L, Diamandis EP. Molecular forms of prostate-specific antigen in
the serum of women with benign and malignant breast diseases. Br J Cancer
1997;76:1087-94.
5. Giai M, Yu H, Roagna R, Ponzone R, Katsaros D, Levesque MA, Diamandis
EP. Prostate-specific antigen in serum of women with breast cancer. Br
J Cancer 1995;72:728-31.
6.
Foekens JA, Diamandis EP, Yu H, Look MP, Meijer-van Gelder ME, van Putten
WLJ, et al. Expression of prostate specific antigen (PSA) correlates with
poor response to tamoxifen therapy in recurrent breast cancer. Br J Cancer
(in press).
7.
Yu H, Giai M, Diamandis EP, Katsaros D, Sutherland DJ, Levesque MA, et
al. Prostate-specific antigen is a new favorable prognostic indicator
for women with breast cancer. Cancer Res 1995;55:2104-10.
8. Yu H, Diamandis EP. Prostate-specific antigen in milk of lactating
women. Clin Chem 1995;41:54-8.
9. Lambert-Messerlian GM, Canick JA, Melegos DN, Diamandis EP. Increased
concentrations of prostate-specific antigen in maternal serum from pregnancies
affected by fetal Down syndrome. Clin Chem 1998;44:205-8.
10.
Tsuyuki D, Grass L, Diamandis EP. Frequent detection of mutations in the
5' flanking region of the prostate-specific antigen gene in female breast
cancer. Eur J Cancer 1997;33:1851-4.
11.
Schuur ER, Henderson GA, Kmetec LA, Miller JD, Lamparski HG, Henderson
DR. Prostate-specific antigen expression is regulated by an upstream enhancer.
J Biol Chem 1996;271:7043-51.
12.
Cleutjens KB, van der Korput HA, van Eekelen CC, van Rooij HC, Faber PW,
Trapman J. An androgen response element in a far upstream enhancer region
is essential for high, androgen-regulated activity of the prostate-specific
antigen promoter. Mol Endocrinol 1997;11:148-61.
13.
Yu H, Diamandis EP, Zarghami N, Grass L. Induction of prostate specific
antigen production by steroids and tamoxifen in breast cancer cell lines.
Breast Cancer Res Treat 1994;32:291-300.
14.
Zarghami N, Grass L, Diamandis EP. Steroid hormone regulation of prostate-specific
antigen gene expression in breast cancer. Br J Cancer 1997;75(4):579-88.
|
 |