|
CEA:
Widely Used and Well Established
Carcinoembryonic
antigen (CEA), identified more than 30 years ago, is used extensively
as a tumor marker for colorectal, gastrointestinal, lung and breast carcinoma,
and for a variety of other tumors that can express elevated serum levels
of the marker. (See Figure 1.) The early work reported on CEA indicated
specificity for cancer, but subsequent research has shown the molecule's
expression in normal colonic mucosa to be similar to that in cancer tissues.
Molecules of CEA have been observed anchored, via mediators of cell signaling,
to cell membranes,1 where they have a
role in intercellular recognition, regulation of the immune response,
and metastasis of colorectal cancer. Although a better understanding of
CEA has been gained in the past three decades, solutions to a number of
outstanding issues might improve the value of this marker in the future.
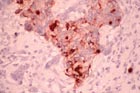 Figure 1. Breast adenocarcinoma section
stained with monoclonal anti-CEA antibody. Breast tumors may exhibit increased
CEA production, which is reflected by elevated serum CEA levels.
Figure 1. Breast adenocarcinoma section
stained with monoclonal anti-CEA antibody. Breast tumors may exhibit increased
CEA production, which is reflected by elevated serum CEA levels.
CEA,
a single-chain polypeptide consisting of 641 amino acids, occurs as heterogeneous,
high molecular weight glycoproteins (175 ‚ 200 kDa), with varying carbohydrate
(45 ‚ 55%) and amino acid content. CEA is a member of a large family of
(up to 36) related cell surface glycoproteins whose genes (ten located
on chromosome 19) can be demonstrated in both normal and diseased colonic
epithelium. These include CEA, nonspecific crossreacting antigen (NCA),
biliary glycoprotein (BGP) and CEA gene family member 2 (CGM2).2
In addition, the structural domains of CEA, NCA and the gammaheavy chain
of the immunoglobulin IgG are very similar; CEA is consequently grouped
in the immunoglobulin gene "superfamily."
In
a recent reference range study of more than 700 individuals from four
European countries, the 95th percentile upper limits of the IMMULITE®
CEA assay for male smokers and nonsmokers were 6.2 and 3.4 microgram/L,
and for female smokers and nonsmokers, 4.9 and 2.5 microgram/L, respectively.3
These results agree with previous studies that found higher reference
values to be associated with smokers, which is an important factor to
consider when interpreting CEA results.
Since
elevated CEA levels can occur in patients with a number of benign diseases,
such as cirrhosis, pulmonary emphysema, rectal polyps, benign breast disease
and ulcerative colitis, and because a number of tumors do not produce
CEA, this marker is not a useful screening tool.
In
colorectal cancer, perioperative CEA levels provide accurate prognostic
information. Elevated postoperative concentrations are used to identify
patients with recurrent disease, particularly patients with hepatic or
pulmonary metastastic disease.4,5 Elevated
preoperative CEA levels correlate inversely with mean time to recurrence
and directly with tumor stage and pathological grading in colorectal cancer
patients. The marker is also used to monitor therapy in patients being
treated by chemotherapy or radiotherapy. Postoperative CEA levels have
been used to identify patients suitable for curative resection (second-look
surgery) when recurrent colorectal cancer has been identified. A recent
study has reported that CEA-driven surgery in selected recurrent colorectal
cancer patients is useful and can produce long-term survivors.6
CEA
has been used in breast cancer patients primarily to monitor disease and
to detect tumor recurrence and metastatic spread. It is accepted that
assays detecting the MUC1 mucin (e.g., BR-MA*, CA15-3) are more sensitive
than CEA and are more often expressed in this disease, but the combined
use of the two types of assay can be the most effective.
A
recent study of breast cancer patients7
showed that both IMMULITE CEA and BR-MA (CA15-3) levels correlated with
stage of disease, indicating that these two markers reflected tumor burden.
Of special note was the fact that serum levels of these markers in Stage
III disease were significantly higher than in patients with early breast
cancer (Stages I and II) or in the normal group. In patients with advanced
breast cancer there was no correlation between the site of metastasis
and elevated tumor marker level, but patients with multiple metastatic
sites had higher marker levels than those with single sites, the marker
levels thus indicating tumor burden. A combination of CEA and CA15-3 has
been reported to improve lead detection time in metastatic disease, more
so than the use of either assay alone.8
In a study of 8,000 breast cancer patients, the combined use of these
two markers provided an easy method of screening for metastases during
patient follow-up.9
A
major study was recently performed at three centers on serial specimens
from patients clinically diagnosed with various cancers, as part of the
clinical verification of the IMMULITE CEA assay.10
The study included 829 specimens from 95 serially monitored patients;
most of the specimens were from colorectal cancer patients. An example
of data from this study appears in Figure 2. The IMMULITE CEA results
accurately reflected the disease status (progression or regression) in
cancer patients in 87 of the 95 cases (91.6%). The eight remaining patients
(8.4%) had CEA results that did not parallel the disease status, indicating
that CEA was not being expressed.
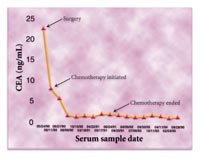 Figure 2. After surgery, CEA levels in
a colorectal cancer patient further declined with initiation of chemotherapy
(06/11/90) and remained low, reflecting the lack of disease following
treatment.
Figure 2. After surgery, CEA levels in
a colorectal cancer patient further declined with initiation of chemotherapy
(06/11/90) and remained low, reflecting the lack of disease following
treatment.
Clinical
practice guidelines for the use of tumor markers in breast and colorectal
cancer have been published by the American Society of Clinical Oncology
(1996).11 For colorectal cancer, CEA
measurement was recommended preoperatively in cases where the level would
influence surgical management. For breast cancer, in the absence of readily
measurable disease, CA15-3 and CEA levels can be used to document treatment
failure.
New
applications of this established tumor marker and improved effectiveness
when used in combination with other markers continue to be reported, ensuring
the enduring value of CEA measurements for cancer patient management.
References
1.
Hammerst–m S, Baranov V, Fr”ngsmyr L, Tjernstr–m A, Zhou G-Q. Further
studies on the possible role of carcinoembryonic antigen (CEA) family
molecules in non-specific defense of colon mucosa. Tumor Biol 1997;18
Suppl 2:9.
2. Matsuoka Y, Kuroki Ma, Kuroki Mo. Cancer specificity problems of CEAã
a review. Tumor Biol 1997;18 Suppl 2;9.
3. DPC data on file.
4. Cooper EH, de Mello JP Jr, Giles GR. Biochemical markers in gastrointestinal
malignancies. Arq Gastroenterol 1989;26(4):131-40.
5. Jessup JM, Thomas P. Carcinoembryonic antigen: function in metastasis
by human colorectal carcinoma. Cancer Metastasis Rev 1989;8:263-80.
6. Lucha PA Jr, Rosen L, Olenwine JA, Reed JF III, Riether RD, Stasik
JJ, et al. Value of carcinoembryonic antigen monitoring in curative surgery
for recurrent colorectal carcinoma. Dis Colon Rectum 1997;40:145-9.
7.
Murray A, Willsher P, Price MR, Dixon AR, Robertson JFR. Evaluation of
the IMMULITE BR-MA and CEA assays and comparison with immunoradiometric
assays for CA15-3 and CEA in breast cancer. Anticancer Res 1997;17:1945-50.
8.
Coveney EC, Geraghty JG, Sherry F, McDermott EW, Fennelly JJ, O¼Higgins
NJ, et al. The clinical value of CEA and CA15-3 in breast cancer management.
Int J Biol Markers 1995;10:35-41.
9. J”ger W, Kr”mer S, Palapelas V and Norbert L. Breast cancer and clinical
utility of CA15-3 and CEA. Scand J Clin Lab Invest 1995;55 Suppl 221;87-92.
10. DPC data on file.
11.
American Society of Clinical Oncology. Clinical practice guidelines for
the use of tumor markers in breast and colorectal cancer. J Clin Oncol
1996;14:2843-77.
*Available outside the US.
|