| |
Probing
the GH–IGF Axis
Laboratory Support for Growth Disorders
by Martin W. Elmlinger, Ph.D.
Assistant Professor in Clinical Chemistry, Endocrinology
University Hospital, Tübingen
Tübingen, Germany
and Werner Kühnel, Ph.D.
Marketing Manager, Scientific Affairs
DPC Biermann, Bad Nauheim, Germany
In the diagnosis and management of conditions associated with growth hormone
(GH) deficiency or excess, the pulsatile secretion of this pituitary hormone
severely limits the information one can expect from a single determination
of GH based on a routine ("random") blood draw. Accordingly, for direct
laboratory assessment of somatotropic function, endocrinologists rely
primarily on provocative testing of GH secretion and on the determination
of spontaneous GH-secretion profiles integrated overnight, or over 24
hours.
Assays for insulin-like
growth factor I (IGF-I) and IGF binding protein 3 (IGFBP-3) have also
become essential laboratory tools in the diagnosis and management of GH-secretion
disorders. These two analytes are secreted at high concentrations from
the liver under the control of GH; they are mediators of the growth-promoting
actions of GH; and they exhibit no problematic diurnal variation. The
three assays thus represent a natural partnership. Within the domain of
growth disorders, assays for IGF-I and IGFBP-3 have a somewhat broader
application than GH assays, being important, not only for diagnosing GH
deficiency (GHD), but also for monitoring therapies with recombinant human
GH (rhGH) in various conditions.
The IGF system is
a complex entity involving two IGFs ("somatomedins") and six IGFBPs, as
well as proteases—the best known being prostate-specific antigen
(PSA)—and other components.1 From
the clinical laboratory's point of view, IGF-I is the more important somatomedin
because circulating levels of IGF-II are less closely regulated by GH;
and IGFBP-3 stands out, not only for circulating at far higher concentrations
than the other five IGFBPs, but also for its greater GH-dependence.
Ordinarily, GH hyper-
and hyposecretion tend to induce corresponding increases and decreases
in the circulating levels of both IGF-I and IGFBP-3, as hepatic production
of both molecules is stimulated by the action of GH on its receptors.
Thus, the inherent relationship between the two poles of the GH-IGF axis
is one of parallelism.
A dramatically different
relationship is seen, however, in the presence of GH receptor defects
(e.g., Laron syndrome), which are quite rare.2
Children born with such a defect exhibit severely impaired linear growth
even though GH circulates at normal to elevated levels: IGF-I levels,
on the other hand, remain decidedly low, as in classic GHD, often below
the 0.1 centile expected for normals of comparable age.3
Growth hormone
excess
Acromegaly, the most common disorder of GH hypersecretion, is often caused
by pituitary tumors. In addition to treatment by conventional means (e.g.,
surgery, radiation, somatostatin analogs), acromegaly is commonly treated
in some countries with GH receptor antagonists: this aims at blunting
the usual impact of elevated GH levels rather than eliminating GH hypersecretion
as such.4 Accordingly, GH measurements could be
of no obvious help in monitoring or optimizing this treatment modality,
whereas it would still make sense to look for normalization of circulating
IGF-I levels.
In the diagnosis of
acromegaly, however, and when this disorder is treated by conventional
means, consensus guidelines from international workshops held in 1999
and 2000 specify roles for GH as well as IGF-I measurements. An IGF-I
level in the normal range can help to exclude the diagnosis and is an
important target for therapy. As for GH, the guidelines call for sequential
measurements: either a 24-hour monitoring of spontaneous levels yielding
an average GH concentration below 2.5 µg/L, or else a seemingly more cost-effective
2-hour oral glucose tolerance test (OGTT) yielding a GH result of 1 µg/L
or less.5
But what if the GH
and IGF-I results conflict? An article published in 2002 by Dimaraki et
al. includes instructive data from a referral center in Ann Arbor, Michigan.6
Figure 1 shows a pair of 24-hour GH profiles for two adults from
this study: one a normal control, the other a patient with active acromegaly
confirmed by laboratory-independent criteria and as yet untreated. Although
both subjects have average GH levels within (conventional) normal limits
for this parameter, the acromegaly patient's IGF-I level exceeds the stated
upper reference limit for the assay used.7
The GH profiles in
Figure 1 exhibit strikingly different topographies: huge swings between
peak and valley, a reflection of normal, intermittent GH secretion, versus
a plateau, indicative of the persistent, "tonic" secretion characteristic
of acromegaly. Statistics like the mean (or median or geometric mean)
fail to discriminate between the normal and acromegaly profiles; instead,
it is the lowest GH concentrations which encapsulate the relevant
difference.
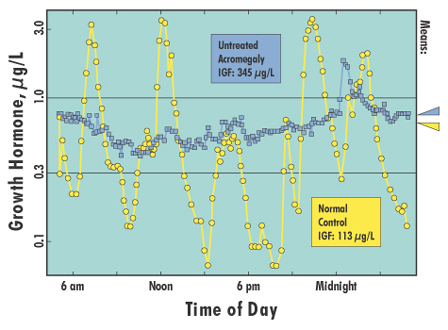 |
| Figure
1. 24-hour GH secretion profiles, sampled at 10-minute intervals,
for two adults. Acromegaly patient, untreated, IGF-I: 345 µg/L (elevated),
GH mean: 0.6 µg/L. Normal control, IGF-I: 113 µg/L (normal), GH mean:
0.7 µg/L. Data from Dimaraki EV, et al. J Clin Endocrinol Metab 2002;87:3537-42.
|
Herein we see one
rationale for the OGTT, as a relatively convenient way to estimate the
very lowest levels expected from a 24-hour profile—rendered less
precise, of course, due to sparse sampling. (From another, purely qualitative
perspective, the point of an OGTT is that failure to suppress soon after
a glucose load suggests an autonomous source of GH production.)
The examples published
by Dimaraki et al. convey several lessons. First, for reliable
measurement of the most significant levels encountered in an OGTT or spontaneous
profile, one needs a GH assay with second- or third-generation sensitivity,
comparable to that of the IMMULITE® and IMMULITE® 2000 Growth Hormone
assays.
Second, for
diagnosis (though not necessarily in treatment follow-up8),
an OGTT cutoff of 1 µg/L, as recommended in the consensus guidelines,
could well be set too high. A value of 0.3 µg/L—closer, presumably,
to the upper reference limit for the lowest OGTT-induced GH levels in
normal subjects—may prove more appropriate for today's nonisotopic,
immunometric assays. Due partly to the specificity of their antibody pairs,
these assays tend to yield numerically lower GH results than RIAs and
first-generation IRMAs on very low patient samples.
Third, in the
diagnosis and follow-up of acromegaly, an elevated IGF-I result—which
can be obtained and checked far more readily than the lowest result of
an OGTT—should not be ignored.
Growth hormone
deficiency
In the diagnosis of GHD, where low concentrations of IGF-I and IGFBP-3
would be expected on the basis of GH hyposecretion, the interpretation
of IGF-I and IGFBP-3 results is complicated by factors competing with
GH secretion for control—most notably nutritional status—and
by nonhepatic IGF production. Immune system status,9
cancers, and even social factors represent additional complications.
Malnutrition may result
in low circulating levels of IGF-I, in spite of normal somatotropic function;10
while obesity may succeed in masking a case of GHD by driving IGF-I levels
higher up into the normal range. It must be said that nutritional status
also affects GH levels, but in opposite directions: GH secretion is typically
decreased in obesity,11 while fasting
increases the frequency and amplitude of GH bursts.
weight.JPG) |
| A
child with Prader-Willi syndrome (PWS), a chromosomal disorder often
associated with short stature as well as a tendency (beginning at
1 to 4 years of age) towards rapid and extreme weight gain. Is GHD
an inherent component of PWS? With nutritional status so often a confounding
factor, it has been difficult to reach a consensus. In the United
States, rhGH was recently approved for use in children with this syndrome,
thus obviating the need to demonstrate GHD on an individual basis
before instituting therapy. Photograph used with permission of the
Prader-Willi Syndrome Association (UK),
www.pwsa-uk.demon.co.uk.
|
IGFs act both as hormones
and as cytokines: they are produced not only in the liver but at many
sites throughout the body. Whereas, in general, one cannot expect local
(i.e., tissue-related) changes in the distribution of IGF and IGFBP levels
to be clearly discernible at the systemic level (i.e., in the circulation),
it is still possible that local production might spill over into the vascular
space just enough to confound the interpretation of low IGF levels.
Nevertheless, in practice,
measurements of IGF-I and IGFBP-3 represent an important step in the work-up
of suspected GHD, especially in children, where it is natural to evaluate
these analytes before embarking on more costly studies involving sequential
GH determinations. The latter remain essential to the diagnosis, however;
and in spite of renewed advocacy for overnight or even 24-hour secretion
profiles in children,12 most endocrinologists
have traditionally sought to document GHD with impaired responses in two
different GH-stimulation tests.13 (Arguably,
the scientific justification for these nonphysiological provocation tests
lies in their ability to yield an index to the peak levels expected
from a spontaneous profile.)
In adults, it is standard
practice to monitor rhGH therapy with periodic IGF-I and IGFBP-3 determinations.
For adult GHD, the prevailing philosophy is one of replacement;
hence the desire to achieve and maintain normal circulating levels of
these analytes, as surrogates for the systemic action of GH, both to minimize
side-effects and because long-term exposure to high GH levels may be a
risk factor for cardiovascular disease and certain epithelial cancers.14
Epidemiological studies have suggested an increased risk for such cancers
in subjects with both high circulating levels of IGF-I and low circulating
levels of IGFBP-3 (hence also an elevated ratio of these two analytes).15
Moreover, with the
gradual appreciation that the optimal rhGH dose depends on age, gender,
steroid levels and other factors, it has become increasingly common to
individualize therapy in adults by starting with a low dose and titrating
upwards, using IGF-I and IGFBP-3 levels for guidance.16
In children, these
two analytes have also been monitored routinely "for assurance of compliance
and safety," but not, until recently, for dose adjustment: generally speaking,
the rhGH dose has been determined solely on weight (or surface area),
as if age, gender and pubertal status were irrelevant.17
In contrast to the
situation for adults, parameters related to a child's height (especially
height velocity) provide an objective basis for measuring the success
of rhGH administration in promoting growth. Furthermore, the list of indications
for rhGH now includes several—e.g., Turner syndrome, SGA (small for
gestational age), and chronic renal failure—where impaired growth
remains at issue, but GHD is not assumed and need not be demonstrated.18
Accordingly, the focus of clinical research has shifted towards ways of
individualizing the rhGH dose, using regression models based on laboratory
results, growth-related parameters and other variables, to achieve optimal
improvements in growth.
Peak levels from overnight
or 24-hour spontaneous GH profiles have so far proved to be the most important
of the laboratory variables in this context, but IGF-I and IGFBP-3 levels
obtained prior to therapy significantly extend the scope of models for
predicting first-year height velocity.19 Whether
serial measurements from routine monitoring with these two analytes will
also contribute towards effective dose adjustment remains an important
open question.
Reference ranges
The within-day stability of circulating IGF-I and IGFBP-3 levels is a
major, though hardly decisive, consideration favoring assays for these
analytes over assays for GH. Published studies have demonstrated essentially
no diurnal variation for IGF-I and IGFBP-3, except during rhGH therapy,
which induces a modest and predictable circadian rhythm.20
(See Figure 2. The difference in IGF-I levels between the two groups
may be due to duration of rhGH treatment, but is more likely due to age;
in any case, the two groups show a comparable degree of within-day variation.)
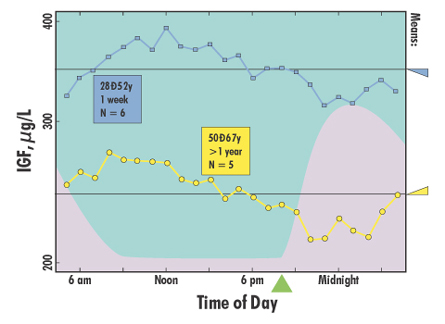 |
| Figure
2. Average 24-hour IGF-I profiles, sampled at 60-minute intervals,
for adults undergoing rhGH therapy. Six patients, 28–52y, after
1 week of treatment. Five patients, 50–67y, treated for 13–40
months. The lavender mound suggests the typical time course for rise
and fall of circulating GH in these patients, with rhGH administered
at 8 pm. Data from Oscarsson J, et al. Clin Endocrinol 1997;46:63-8.
|
This has immediate
implications, not only for the comparative number of measurements required
for reliable assessment of GH versus IGF-I and IGFBP-3, but also for the
possibility of obtaining definitive interpretative frames of reference
for these analytes. To establish normal values for IGF-I and IGFBP-3,
only a single "random" determination is required for each subject in the
reference group, whereas this is not true for GH, due to the variety of
testing formats, as well as the need for multiple determinations.
Taking not just the
agents, but also dosing criteria, timing, patient preparation and other
significant features into consideration, GH provocation tests have been
applied in an astounding number of combinations. There seems little hope
for standardization; and even less for the rigorous, scientific determination
of assay-specific reference values and cutoffs. It is difficult to imagine
an internal review board approving normal range studies of a statistically
adequate size for GH provocation tests, especially in children, given
that the tests are nonphysiological and often involve some risk.
Accordingly, except
possibly for overnight studies of spontaneous GH secretion—which
are not open to this objection, though they are similarly arduous and
costly—one must expect that cutoffs for GH tests will continue to
rely on "traditional" values or on studies with very limited N-size that
fail to take potentially significant variables into account—particularly
age, gender, and pubertal status (Tanner stage) or steroid levels.
The same variables
are relevant, of course, to IGF-I and IGFBP-3 as well. This means that
establishing reference values entails a complex, multivariate study. Moreover,
in this domain, it is expected that such a study will conform to the rigor
and methodology associated with the development of growth standards for
weight, height, height velocity and related parameters, rather than the
less demanding IFCC/NCCLS guidelines for minimalist, two-point characterizations
of reference distributions (e.g., in terms of central 95% reference intervals)
which are still accepted as adequate in other parts of clinical chemistry.21
In developing reference
values for IGF-I and IGFBP-3, it is also important to determine their
ratio, which yields a valuable cross-check on the integrity of the data.
In many applications, it is common practice to measure IGFBP-3 along with
IGF-I for the same reason, that is, to use the former as an "internal
control" for the latter. Furthermore—analogous to the use of T4/TBG
and testosterone/SHBG ratios in other contexts—the IGF-I / IGFBP-3
ratio serves as an index of circulating free IGF-I.22
Some of the analyses
of reference values essential to the routine evaluation of growth disorders
are illustrated in the accompanying figures, which show the expected distribution
of IGF-I and IGFBP-3 and their ratio as a function of age (Figure 3) and
Tanner Stage (Figure 4), obtained in a major recent study of children
and adults in good health and of normal height.23
Conclusions
While there are other promising applications for circulating GH, IGF-I
and IGFBP-3 measurements, the best established relate to disorders of
growth.24 Laboratory results on their
own cannot settle the issues which arise for endocrinologists, who must
take far more than immunoassay results into consideration. But the laboratory's
contribution can be optimized by ensuring that the assays used for these
three analytes meet certain basic criteria.
For GH, a highly sensitive,
specific immunometric assay is essential: automation and small sample
volume are desirable features given that sequential determinations (based
on provocation tests and/or spontaneous profiles) are the norm.
For IGF-I and IGFBP-3,
and for their ratio, a principal requirement is rigorous, detailed, assay-specific
characterizations of the normal reference distribution as a function of
age, sex (where appropriate) and Tanner stage.
Notes
1. IGFs, IGFBPs, proteases:
[Ran97], [Juu98], [Fer99], [Gri00], [LeR01], [Fir02]. See also [R&C02],
an excellent general reference, in Sperling's Pediatric Endocrinology.
2.
Laron syndrome, GH resistance: [Lar99], [Lop00], [Ros01]. GHD as a subtype
of IGFD: [Ros94b], [Ros96], [R&C02]. The Ecuador story: [Ros90], [Ros94a].
3.
The 0.1 centile for IGF-I: [Blu94], [Blu00].
4.
GH receptor antagonists: [Kop02], [Tra02a].
5.
Consensus guidelines, acromegaly: [Giu00], [Mel02b]. Note that the 2002
guidelines do not mention spontaneous GH profiles. See also [Mel99], [Mel02a],
[Giu03].
6.
Dimaraki study, Figure 1: [Dim02]. See notes 6 and 7.
7.
Apropos of [Dim02]: [Tra02b], [Fre02], [Giu03], [Vie03].
8.
Not necessarily in treatment follow-up: [Cos02].
9.
Immune system status: [Blu00].
10.
Nutritional status: [Thi94], [Bar02], [Doh02], [R&C02], [Hol03]. Monitoring
refeeding: [Cle85].
11.
Prader-Willi syndrome, obesity: [Eih98], [Eih00], [Eih01], [Bar02], [Car02].
12.
Spontaneous GH profiles, pro and con: [Kri01]; [Guy99], [Rog03]. See also
note 19.
13.
GH provocation testing. [Juu97b], [GRS98], [Sag98], [Sha98], [GRS00],
[Siz01], [Jor03]. Clinical practice audits: [Wya95], [Juu02a].
14.
Monitoring rhGH therapy in adults: [Juu99a], [GRS01], [Dra01], [Mur02].
15.
Cancer risk: [Gri00], [Coh01], [GRS01], [R&C02], [Bar02], [Zha03]. See
especially: [Coh00]. Cancer diagnosis: [Cut99], [Sta01], [Ism02], [Sco03].
16.
Individualizing rhGH therapy in adults: [Dra98], [Mur00], [Van01], [Mol02],
[Mur02], [Sim02].
17.
Monitoring rhGH therapy in children: [Dra98], [GRS01], [Lee01], [Ran01],
[Juu02a], [Spe02].
18.
Use of rhGH for conditions other than confirmed GHD: [AAP97], [AACE98],
[Guy99], [Juu99b], [R&C02], [Ran03].
19.
Individualizing rhGH therapy in children: [Wet00], [Lee01], [Ber02], [Coh02].
Prediction models: [Alb00], [Kri01], [Ran01], [Sch01], [Kri02], [Ran03].
20.
Diurnal variation: [Dra01], [Gel99], [Heu99], [Juu99a]. Figure 2: [Osc97].
21.
Growth standards, age-related reference ranges: [Ran00], [Sib00], [Juu01],
[Lof01], [R&C02], [Elm03].
22.
IGF-I/IGFBP-3 ratio and "free" IGF-I: [Juu97a] [Heu99], [Blu00], [Ban01],
[Fry01]. See also note 15.
23.
Tübingen IGF-I reference range study, Figures 3 and 4: [Wen02], [Elm03],
[Kuh03].
24.
On the horizon: [Geu98], [Ros99], [Bar00], [Ses00], [Bar02], [Bje02],
[Fry02], [Juu02b], [Daw03], [Gio03], [Hol03].
References
[AACE98]
American Association of Clinical Endocrinologists (www.aace.com/pub/ep/).
AACE clinical practice guidelines for growth hormone use in adults and
children. Endocrine Practice 1998;4(3):166-73.
[AAP97]
American Academy of Pediatrics, Committee on Drugs and Committee on Bioethics
(www.aap.org). Considerations related to the use of recombinant human
growth hormone in children. Pediatrics 1997;99:122-9. UI=97143337.
[Alb00] Albertsson Wikland K, Kristrom
B, Rosberg S, Svensson B, Nierop AF. Validated multivariate models predicting
the growth response to GH treatment in individual short children with
a broad range in GH secretion capacities. Pediatr Res 2000;48:475-84.
UI=20461410.
[Ban01] Bang P, Ahlsen M, Berg U, Carlsson-Skwirut
C. Free insulin-like growth factor I; are we hunting a ghost? Horm Res
2001;55 Suppl 2:84-93. UI=21541387.
[Bar00] Barkan AL, Clemmons DR, Molitch
ME, Stewart PM, Young WF Jr. Growth hormone therapy for hypopituitary
adults; time for re-appraisal. Trends Endocrinol Metab 2000;11:238-45.
UI=21120134.
[Bar02] Barnard RJ, Aronson WJ, Tymchuk
CN, Ngo TH. Prostate cancer; another aspect of the insulin-resistance
syndrome? Obes Rev 2002;3:303-8. UI=22346790.
[Ber02] Bercu BB. Titration of growth hormone
dose using insulin-like growth factor-1 measurements; is it feasible in
children? J Pediatr 2002;141:601-5. UI=22297381.
[Bje02] Bjerregård Sneppen S, Steensgaard-Hansen
F, Feldt-Rasmussen U. Cardiac effects of low-dose growth hormone replacement
therapy in growth hormone-deficient adults; an 18-month randomised, placebo-controlled,
double-blind study. Horm Res 2002;58:21-9. UI=22159868.
[Blu94] Blum WF, Cotterill AM, Postel-Vinay
MC, Ranke MB, Savage MO, Wilton P. Improvement of diagnostic criteria
in growth hormone insensitivity syndrome; solutions and pitfalls. Acta
Paediatr Suppl 1994;399:117-24. UI=95036902.
[Blu00] Blum WF. Insulin-like growth factors
(IGF) and IGF-binding proteins; their use for diagnosis of growth hormone
deficiency. In: Juul A, Jørgensen JOL. Growth hormone in adults; physiological
and clinical aspects. 2nd ed. Cambridge: Cambridge University Press; 2000.
p. 54-86.
[Car02] Carrel AL, Myers SE, Whitman BY,
Allen DB. Benefits of long-term GH therapy in Prader-Willi syndrome; a
4-year study. J Clin Endocrinol Metab 2002;87:1581-5. UI=21929862.
[Cle85] Clemmons DR, Underwood LE, Dickerson
RN, Brown RO, Hak LJ, MacPhee RD, Heizer WD. Use of plasma somatomedin-C/insulin-like
growth factor I measurements to monitor the response to nutritional repletion
in malnourished patients. Am J Clin Nutr 1985;41:191-8. UI=85119105.
[Coh00] Cohen P, Clemmons DR, Rosenfeld
RG. Does the GH-IGF axis play a role in cancer pathogenesis? Growth Horm
IGF Res 2000;10:297-305. UI=21112234.
[Coh01] Cohen P. Clinical implications
of the IGF-cancer connection. Growth Horm IGF Res 2001;11:336-8. UI=21912084.
[Coh02] Cohen P, Bright GM, Rogol AD, Kappelgaard
AM, Rosenfeld RG. Effects of dose and gender on the growth and growth
factor response to GH in GH-deficient children; implications for efficacy
and safety. J Clin Endocrinol Metab 2002 Jan;87:90-8. UI=21648788.
[Cos02] Costa AC, Rossi A, Martinelli CE
Jr, Machado HR, Moreira AC. Assessment of disease activity in treated
acromegalic patients using a sensitive GH assay; should we achieve strict
normal GH levels for a biochemical cure? J Clin Endocrinol Metab 2002;87:3142-7.
UI=22103230.
[Cut99] Cutting CW, Hunt C, Nisbet JA,
Bland JM, Dalgleish AG, Kirby RS. Serum insulin-like growth factor-1 is
not a useful marker of prostate cancer. BJU Int 1999;83:996-9. UI=99296791.
[Daw03] Dawson-Hughes B. Interaction of
dietary calcium and protein in bone health in humans. J Nutr 2003;133:852s-854s.
UI=22499818.
[Dim02] Dimaraki EV, Jaffe CA, DeMott-Friberg
R, Chandler WF, Barkan AL. Acromegaly with apparently normal GH secretion;
implications for diagnosis and follow-up. J Clin Endocrinol Metab 2002;87:3537-42.
UI=22151237.
[Doh02] Doherty CP, Crofton PM, Sarkar
MA, Shakur MS, Wade JC, Kelnar CJ, Elmlinger MW, Ranke MB, Cutting WA.
Malnutrition, zinc supplementation and catch-up growth: changes in insulin-like
growth factor I, its binding proteins, bone formation and collagen turnover.
Clin Endocrinol (Oxf) 2002;57:391-9. UI=22190812.
[Dra98] Drake WM, Coyte D, Camacho-Hubner
C, Jivanji NM, Kaltsas G, Wood DF, Trainer PJ, Grossman AB, Besser GM,
Monson JP. Optimizing growth hormone replacement therapy by dose titration
in hypopituitary adults. J Clin Endocrinol Metab 1998;83:3913-9. UI=99029605.
[Dra01] Drake WM, Howell SJ, Monson JP,
Shalet SM. Optimizing GH therapy in adults and children. Endocr Rev 2001;22:425-50.
UI=21385006.
[Eih98] Eiholzer U, Stutz K, Weinmann C,
Torresani T, Molinari L, Prader A. Low insulin, IGF-I and IGFBP-3 levels
in children with Prader-Labhart-Willi syndrome. Eur J Pediatr 1998;157:890-3.
UI=99051095.
[Eih00] Eiholzer U, Bachmann S, l'Allemand
D. Is there growth hormone deficiency in Prader-Willi syndrome? Six arguments
to support the presence of hypothalamic growth hormone deficiency in Prader-Willi
syndrome. Horm Res 2000;53 Suppl 3:44-52. UI=20428617.
[Eih01] Eiholzer U. Prader-Willi syndrome:
effects of human growth hormone treatment. Basel: Karger; 2001.
[Elm03] Elmlinger MW, Kühnel W, et al.
Adaptive local regression methods for age-related reference range analysis
applied to IMMULITE IGF-I and IGFBP-3 data. Clin Chem 2003;49(6 Suppl).
[Fer99] Ferry RJ Jr, Cerri RW, Cohen P.
Insulin-like growth factor binding proteins: new proteins, new functions.
Horm Res 1999;51:53-67. UI=99282542.
[Fir02] Firth SM, Baxter RC. Cellular actions
of the insulin-like growth factor binding proteins. Endocr Rev 2002;23:824-54.
UI=22354142.
[Fre02] Freda PU. Somatostatin analogs
in acromegaly. J Clin Endocrinol Metab 2002;87:3013-8. UI=22103208.
[Fry01] Frystyk J, Ivarsen P, Stoving RK,
Dall R, Bek T, Hagen C, Orskov H. Determination of free insulin-like growth
factor-I in human serum: comparison of ultrafiltration and direct immunoradiometric
assay. Growth Horm IGF Res 2001;11:117-27. UI=21365720.
[Fry02] Frystyk J, Ledet T, Moller N, Flyvbjerg
A, Orskov H. Cardiovascular disease and insulin-like growth factor I.
Circulation 2002;106:893-5. UI=22174357.
[Gel99] Gelander L, Blum WF, Larsson L,
Rosberg S, Albertsson Wikland K. Monthly measurements of insulin-like
growth factor I (IGF-I) and IGF-binding protein-3 in healthy prepubertal
children; characterization and relationship with growth; the 1-year growth
study. Pediatr Res 1999;45:377-83. UI=99186519.
[Geu98] Geusens P, Bouillon R, Broos P,
Rosen DM, Adams S, Sanders M, Raus J, Boonen S. Musculoskeletal effects
of recombinant human insulin-like growth factor-I (rhIGF-I) / IGF binding
protein-3 (IGFBP-3) in hip fracture patients; results from a double blind
placebo-controlled phase II study (Abstract). Bone 1998;23(5):s157.
[Gio03] Giovannucci E, Pollak M, Liu Y,
Platz EA, Majeed N, Rimm EB, Willett WC. Nutritional predictors of insulin-like
growth factor I and their relationships to cancer in men. Cancer Epidemiol
Biomarkers Prev 2003;12:84-9. UI=22468983.
[Giu00] Giustina A, Barkan A, Casanueva
FF, Cavagnini F, Frohman L, Ho K, Veldhuis J, Wass J, Von Werder K, Melmed
S. Criteria for cure of acromegaly; a consensus statement. J Clin Endocrinol
Metab 2000;85:526-9. UI=20152708.
[Giu03] Giustina A, Melmed S. Acromegaly
consensus; the next steps. J Clin Endocrinol Metab 2003;88:1913-4.
[Gri00] Grimberg A, Cohen P. Role of insulin-like
growth factors and their binding proteins in growth control and carcinogenesis.
J Cell Physiol 2000;183:1-9. UI=20164637.
[GRS98] Consensus guidelines for the diagnosis
and treatment of adults with growth hormone deficiency; summary statement
of the Growth Hormone Research Society Workshop on Adult Growth Hormone
Deficiency. J Clin Endocrinol Metab 1998;83:379-81. UI=98128674.
[GRS00] Consensus guidelines for the diagnosis
and treatment of growth hormone (GH) deficiency in childhood and adolescence;
summary statement of the GH Research Society. J Clin Endocrinol Metab
2000;85:3990-3. UI=20544528.
[GRS01] Critical evaluation of the safety
of recombinant human growth hormone administration; statement from the
Growth Hormone Research Society. J Clin Endocrinol Metab 2001;86:1868-70.
UI=21242758.
[Guy99] Guyda HJ. Four decades of growth
hormone therapy for short children; what have we achieved? J Clin Endocrinol
Metab 1999;84:4307-16. UI=20066663.
[Heu99] Heuck C, Skjaerbaek C, Orskov H,
Wolthers OD. Circadian variation in serum free ultrafiltrable insulin-like
growth factor I concentrations in healthy children. Pediatr Res 1999;45:733-6.
UI=99248655.
[Hol03] Holt RI, Simpson HL, Sonksen PH.
The role of the growth hormone-insulin-like growth factor axis in glucose
homeostasis. Diabet Med 2003;20:3-15. UI=22407634.
[Ism02] Ismail A H, Pollak M, Behlouli
H, Tanguay S, Begin LR, Aprikian AG. Insulin-like growth factor-1 and
insulin-like growth factor binding protein-3 for prostate cancer detection
in patients undergoing prostate biopsy. J Urol 2002 Dec;168:2426-30. UI=22329292.
[Jor03] Jorgensen JO, Christiansen JS.
Testing for growth hormone deficiency in adults; how to respond? Clin
Endocrinol (Oxf) 2003;58:18-9. UI=22407425.
[Juu97a] Juul A, Holm K, Kastrup KW, Pedersen
SA, Michaelsen KF, Scheike T, Rasmussen S, Muller J, Skakkebaek NE. Free
insulin-like growth factor I serum levels in 1430 healthy children and
adults, and its diagnostic value in patients suspected of growth hormone
deficiency. J Clin Endocrinol Metab 1997;82:2497-502. UI=97397263.
[Juu97b] Juul A, Kastrup KW, Pedersen SA,
Skakkebaek NE. Growth hormone (GH) provocative retesting of 108 young
adults with childhood-onset GH deficiency and the diagnostic value of
insulin-like growth factor I (IGF-I) and IGF-binding protein-3. J Clin
Endocrinol Metab 1997;82:1195-201. UI=97255249.
[Juu98] Juul A, Moller S, Mosfeldt-Laursen
E, Rasmussen MH, Scheike T, Pedersen SA, Kastrup KW, Yu H, Mistry J, Rasmussen
S, Muller J, Henriksen J, Skakkebaek NE. The acid-labile subunit of human
ternary insulin-like growth factor binding protein complex in serum: hepatosplanchnic
release, diurnal variation, circulating concentrations in healthy subjects,
and diagnostic use in patients with growth hormone deficiency. J Clin
Endocrinol Metab 1998;83:4408-15. UI=99067111.
[Juu99a] Juul A. Determination of insulin-like
growth factor-I in the monitoring of growth hormone treatment with respect
to efficacy of treatment and side effects: should potential risks of cardiovascular
disease and cancer be considered? Horm Res 1999;51 Suppl 3:141-8. UI=20062749.
[Juu99b] Juul A, Bernasconi S, Chatelain
P, Hindmarsh P, Hochberg Z, Hokken-Koelega A, de Muinck Keizer-Schrama
SM, Kiess W, Oberfield S, Parks J, Strasburger CJ, Volta C, Westphal O,
Skakkebaek NE. Diagnosis of growth hormone (GH) deficiency and the use
of GH in children with growth disorders. Horm Res 1999;51:284-99. UI=20108881.
[Juu01] Juul A. Determination of insulin-like
growth factor I in children; normal values and clinical use. Horm Res
2001;55 Suppl 2:94-9. UI=21541388.
[Juu02a] Juul A, Bernasconi S, Clayton
PE, Kiess W, DeMuinck-Keizer Schrama S. European audit of current practice
in diagnosis and treatment of childhood growth hormone deficiency. Horm
Res 2002;58:233-41. UI=22289147.
[Juu02b] Juul A, Scheike T, Davidsen M,
Gyllenborg J, Jorgensen T. Low serum insulin-like growth factor I is associated
with increased risk of ischemic heart disease; a population-based case-control
study. Circulation 2002;106:939-44. UI=22174366.
[Kop02] Kopchick JJ, Parkinson C, Stevens
EC, Trainer PJ. Growth hormone receptor antagonists; discovery, development,
and use in patients with acromegaly. Endocr Rev 2002;23:623-46. UI=22259838.
[Kri01] Kristrom B, Lofqvist C, Rosberg
S, Albertsson Wikland K. Effect of spontaneous GH secretion and the GH
sampling period on the accuracy of models for predicting growth responses
to GH treatment. J Clin Endocrinol Metab 2001;86:4963-4. UI=21486610.
[Kri02] Kristrom B, Albertsson Wikland
K. Growth prediction models, concept and use. Horm Res 2002;57 Suppl 2:66-70.
UI=22060637.
[Kuh03] Kühnel W. Growth diagnostics; clinical
relevance of growth hormone, IGF-I and IGFBP-3. Los Angeles: Diagnostic
Products Corp, 2003 (in press), Technical report ZBxxx-A. English translation,
with updated graphs and numeric tables, of: Kühnel W. Wachstumsdiagnostik;
klinische Relevanz von hGH, IGF-I und IGFBP-3. News & Views (DPC Biermann)
2002 Oct: 1-18.
[Lar99] Laron Z. The essential role of
IGF-I: lessons from the long-term study and treatment of children and
adults with Laron syndrome. J Clin Endocrinol Metab 1999;84:4397-404.
UI=20066677.
[Lee01] Lee KW, Cohen P. Individualizing
growth hormone dosing in children. Horm Res 2001;56 Suppl 1:29-34. UI=21646292.
[LeR01] Le Roith D, Scavo L, Butler A.
What is the role of circulating IGF-I? Trends Endocrinol Metab 2001;12:48-52.
UI=21129167.
[Lof01] Lofqvist C, Andersson E, Gelander
L, Rosberg S, Blum WF, Albertsson Wikland K. Reference values for IGF-I
throughout childhood and adolescence; a model that accounts simultaneously
for the effect of gender, age, and puberty. J Clin Endocrinol Metab 2001;86:5870-6.
UI=21601920.
[Lop00] Lopez-Bermejo A, Buckway CK, Rosenfeld
RG. Genetic defects of the growth hormone-insulin-like growth factor axis.
Trends Endocrinol Metab 2000;11:39-49. UI=21121726.
[Mel99] Melmed S. Confusion in clinical
laboratory GH and IGF-I reports. Pituitary 1999;2:171-2. UI=20533640.
[Mel02a] Melmed S. Acromegaly. In: Melmed
S, editor. The pituitary. 2nd ed. Oxford: Blackwell; 2002. p. 419-54.
[Mel02b] Melmed S, Casanueva FF, Cavagnini
F, Chanson P, Frohman L, Grossman A, Ho K, Kleinberg D, Lamberts S, Laws
E, Lombardi G, Vance ML, Werder KV, Wass J, Giustina A. Guidelines for
acromegaly management. J Clin Endocrinol Metab 2002;87:4054-8. UI=22202370.
[Mol02] Molitch ME. Diagnosis of GH deficiency
in adults; how good do the criteria need to be? J Clin Endocrinol Metab
2002;87:473-6. UI=21826508.
[Mur00] Murray RD, Howell SJ, Lissett CA,
Shalet SM. Pre-treatment IGF-I level is the major determinant of GH dosage
in adult GH deficiency. Clin Endocrinol (Oxf) 2000;52:537-42. UI=20252983.
[Mur02] Murray RD, Shalet SM. Adult growth
hormone replacement: lessons learned and future direction. J Clin Endocrinol
Metab 2002;87:4427-8. UI=22250911.
[Osc97] Oscarsson J, Johannsson G, Johansson
JO, Lundberg PA, Lindstedt G, Bengtsson BA. Diurnal variation in serum
insulin-like growth factor (IGF)-I and IGF binding protein-3 concentrations
during daily subcutaneous injections of recombinant human growth hormone
in GH-deficient adults. Clin Endocrinol (Oxf) 1997;46:63-8. UI=97212741.
[R&C02] Rosenfeld RG, Cohen P. Disorders
of growth hormone / insulin-like growth factor secretion and action. In:
Sperling MA, editor. Pediatric endocrinology. 2nd ed. New York: Saunders;
2002. p. 211-88.
[Ran97] Ranke MB, Elmlinger M. Functional
role of insulin-like growth factor binding proteins. Horm Res 1997;48
Suppl 4:9-15. UI=98011526.
[Ran00] Ranke MB, Schweizer R, Elmlinger
MW, Weber K, Binder G, Schwarze CP, Wollmann HA. Significance of basal
IGF-I, IGFBP-3 and IGFBP-2 measurements in the diagnostics of short stature
in children. Horm Res 2000;54:60-8. UI=21150195.
[Ran01] Ranke MB, Schweizer R, Elmlinger
MW, Weber K, Binder G, Schwarze CP, Wollmann HA. Relevance of IGF-I, IGFBP-3,
and IGFBP-2 measurements during GH treatment of GH-deficient and non-GH-deficient
children and adolescents. Horm Res 2001;55(3):115-24. UI=21433856.
[Ran03] Ranke MB, Lindberg A, Cowell CT,
Albertsson Wikland K, Reiter EO, Wilton P, Price DA. Prediction of response
to growth hormone treatment in short children born small for gestational
age; analysis of data from KIGS (Pharmacia International Growth Database).
J Clin Endocrinol Metab 2003;88:125-31. UI=22407873.
[Rog03] Rogol AD, Blethen SL, Sy JP, Veldhuis
JD. Do growth hormone (GH) serial sampling, insulin-like growth factor-I
(IGF-I) or auxological measurements have an advantage over GH stimulation
testing in predicting the linear growth response to GH therapy? Clin Endocrinol
(Oxf) 2003;58:229-37. UI=22469081.
[Ros90] Rosenbloom AL, Guevara Aguirre
J, Rosenfeld RG, Fielder PJ. The little women of Loja; growth hormone-receptor
deficiency in an inbred population of southern Ecuador. N Engl J Med 1990
15;323:1367-74. UI=91042841.
[Ros94a] Rosenbloom AL, Guevara-Aguirre
J. PRISMATIC case: bienvenidos a mi tierra de soledad: from poetry to
molecular biology in southern Ecuador. J Clin Endocrinol Metab 1994;79:695-702.
UI=94358065.
[Ros94b] Rosenfeld RG, Rosenbloom AL, Guevara-Aguirre
J. Growth hormone (GH) insensitivity due to primary GH receptor deficiency.
Endocr Rev 1994;15:369-90. UI=94357161.
[Ros96] Rosenfeld RG. Biochemical diagnostic
strategies in the evaluation of short stature; the diagnosis of insulin-like
growth factor deficiency. Horm Res 1996;46:170-3. UI=97107891.
[Ros99] Rosen CJ, Pollak M. Circulating
IGF-I: new perspectives for a new century. Trends Endocrinol Metab 1999;10:136-41.
PMID=10322407.
[Ros01] Rosenfeld RG, Buckway CK. Growth
hormone insensitivity syndromes; lessons learned and opportunities missed.
Horm Res 2001;55 Suppl 2:36-9. UI=21541377.
[Sag98] Saggese G, Ranke MB, Saenger P,
Rosenfeld RG, Tanaka T, Chaussain JL, Savage MO. Diagnosis and treatment
of growth hormone deficiency in children and adolescents; towards a consensus.
Horm Res 1998;50:320-40. UI=99141172.
[Sch01] Schonau E, Westermann F, Rauch
F, Stabrey A, Wassmer G, Keller E, Bramswig J, Blum WF. A new and accurate
prediction model for growth response to growth hormone treatment in children
with growth hormone deficiency. Eur J Endocrinol 2001;144:13-20. UI=21097211.
[Sco03] Scorilas A, Plebani M, Mazza S,
Basso D, Soosaipillai AR, Katsaros N, Pagano F, Diamandis EP. Serum human
glandular kallikrein (hK2) and insulin-like growth factor 1 (IGF-1) improve
the discrimination between prostate cancer and benign prostatic hyperplasia
in combination with total and percent free PSA. Prostate 2003;54:220-9.
UI=22406355.
[Ses00] Sesmilo G, Biller BM, Llevadot
J, Hayden D, Hanson G, Rifai N, Klibanski A. Effects of growth hormone
administration on inflammatory and other cardiovascular risk markers in
men with growth hormone deficiency; a randomized, controlled clinical
trial. Ann Intern Med 2000;133:111-22. UI=20340410.
[Sha98] Shalet SM, Toogood A, Rahim A,
Brennan BM. The diagnosis of growth hormone deficiency in children and
adults. Endocr Rev 1998;19:203-23. UI=98231556.
[Sib00] Sibley PEC. Reference range analysis;
lessons from PSA. News & Views (DPC) 2000 Winter;14(1):9-12. Available
at DPC's Web site, www.dpcweb.com, under Technical Documents, News & Views,
Winter 2000. Later reissued in black & white as technical report ZB204-B.
Los Angeles: Diagnostic Products Corp, 2001.
[Sim02] Simpson H, Savine R, Sonksen P,
Bengtsson BA, Carlsson L, Christiansen JS, Clemmons D, Cohen P, Hintz
R, Ho K, Mullis P, Robinson I, Strasburger C, Tanaka T, Thorner M. Growth
hormone replacement therapy for adults; into the new millennium. Growth
Horm IGF Res 2002;12:1-33. UI=22123410.
[Siz01] Sizonenko PC, Clayton PE, Cohen
P, Hintz RL, Tanaka T, Laron Z. Diagnosis and management of growth hormone
deficiency in childhood and adolescence; part 1, diagnosis of growth hormone
deficiency. Growth Horm IGF Res 2001;11:137-65. UI=21601159.
[Spe02] Sperling MA, Saenger PH, Ray Hintz,
Tom Wilson, Rose SR. Growth hormone treatment and neoplasia; coincidence
or consequence? J Clin Endocrinol Metab 2002;87:5351-2. UI=22354336.
[Sta01] Stattin P, Stenman UH, Riboli E,
Hallmans G, Kaaks R. Ratios of IGF-I, IGF binding protein-3, and prostate-specific
antigen in prostate cancer detection. J Clin Endocrinol Metab 2001;86:5745-8.
UI=21601898.
[Thi94] Thissen JP, Ketelslegers JM, Underwood
LE. Nutritional regulation of the insulin-like growth factors. Endocr
Rev 1994;15:80-101. UI=94208469.
[Tra02a] Trainer PJ. A giant's step forward.
Clin Endocrinol (Oxf) 2002;56:423-5. UI=21963659.
[Tra02b] Trainer PJ. Acromegaly; consensus,
what consensus? J Clin Endocrinol Metab 2002;87:3534-6. UI=22151236.
[Van01] Vance ML. From fixed to individual
dosing in growth hormone-deficient adults. Horm Res 2001;56 Suppl 1:35-7.
UI=21646293.
[Vie03] Vierhapper H, Heinze G, Gessl A,
Exner M, Bieglmayr C. Use of the oral glucose tolerance test to define
remission in acromegaly. Metabolism 2003;52:181-5. UI=22489508.
[Wen02] Wen J, Bostanian Z, Lei J-D, Whitters
E, Pregger K, Sustarsic D, Unver E, Kühnel W, Elmlinger MW, El Shami AS.
Automated chemiluminescent immunoassays of insulin-like growth factor-I
and insulin-like growth factor binding protein 3 on the IMMULITE analyzer.
Poster presentation P-12, Symposium on functional role of IGFBPs, Tübingen
2003. Horm Res 2002;58:263.
[Wet00] Wetterau L, Cohen P. New paradigms
for growth hormone therapy in children. Horm Res 2000;53 Suppl 3:31-6.
UI=20428614.
[Wya95] Wyatt DT, Mark D, Slyper A. Survey
of growth hormone treatment practices by 251 pediatric endocrinologists.
J Clin Endocrinol Metab 1995;80:3292-7. UI=96064797.
[Zha03] Zhao H, Grossman HB, Spitz MR,
Lerner SP, Zhang K, Wu X. Plasma levels of insulin-like growth factor-1
and binding protein-3, and their association with bladder cancer risk.
J Urol 2003;169:714-7. UI=22432492.
PubMed "unique
identifiers" (89 UI):
22499818 22489508
22469081 22468983 22432492 22407873 22407634 22407425 22406355 22354336
22354142 22346790 22329292 22297381 22289147 22259838 22250911 22202370
22190812 22174366 22174357 22159868 22151237 22151236 22123410 22103230
22103208 22060637 21963659 21929862 21912084 21826508 21648788 21646293
21646292 21601920 21601898 21601159 21541388 21541387 21541377 21486610
21433856 21385006 21365720 21242758 21150195 21129167 21121726 21120134
21112234 21097211 20544528 20533640 20461410 20428617 20428614 20340410
20252983 20164637 20152708 20108881 20066677 20066663 20062749 10322407
99296791 99282542 99248655 99186519 99141172 99067111 99051095 99029605
98231556 98128674 98011526 97397263 97255249 97212741 97143337 97107891
96064797 95036902 94358065 94357161 94208469 91042841 85119105
|
