  |
Homocysteine—
To Test and to Treat
Homocysteine is receiving
a lot of attention these days as a new risk factor
for a variety of diseases. During the past 10 years, the vast majority
of nearly 100 case-control retrospective and prospective studies have
shown that
homocysteine is a strong independent risk factor for coronary artery disease,
cerebrovascular disease and peripheral vascular disease. More recent studies
now implicate homocysteine as a risk factor for neural tube defects in
newborns and for cognitive dysfunction disorders such as vascular dementia
and Alzheimer's disease.
What is homocysteine,
where does it come from, and how is it metabolized? Homocysteine is
a normal metabolite of the essential amino acid methionine (Figure 1).
Structurally, it closely resembles methionine and cysteine; all three
amino acids contain sulfur. They are metabolically linked to each other
as shown in Figure 2. Since foods contain little or no free homocysteine,
nearly all of the homocysteine in the body is derived from methionine
in animal and plant proteins.
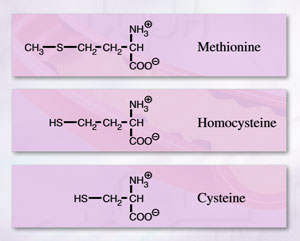
Figure
1. Structure of methione, homocysteine and cysteine.
Homocysteine
metabolism is driven by several B-complex cofactors. Folate and vitamins
B2, B6 and B12 are used in the remethylation pathway; and vitamin B6 is
used in the transsulfuration pathway (Figure 2). Deficiencies of folate,
vitamin B6 or vitamin B12 can lead to impaired homocysteine metabolism
and hyper- homocysteinemia. In addition, mutations in the genes coding
for methyl- enetetrahydrofolate reductase (MTHFR), methionine synthase
(MS) and cystathionine b-synthase (CBS) may
also produce hyperhomocysteinemia. Subjects who inherit two identical
defective alleles may have little or no enzyme activity (e.g., for CBS).
This can result in severe hyperhomocysteinemia and the rare disease known
as homocystinuria. Without treatment, most affected individuals will experience
a cardiovascular event before age 30.
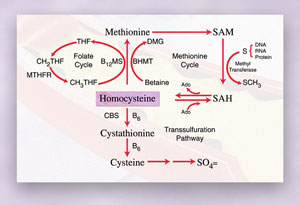
Figure
2. Major pathways of homocysteine metabolism in the liver and kidneys.
Homocysteine is generated in a cycle through S-adenosylmethionine (SAM)
and S-adenosylhomocysteine (SAH). Remethylation of homocysteine back to
methionine is carried out by vitamin B12-dependent methionine synthase
(B12MS) and betaine-homocysteine methyltransferase (BHMT). Homocysteine
is also converted to cysteine through the transsulfuration pathway, initiated
by B6-dependent cystathionine b-synthase (CBS).
The folate cycle generates 5-methyltetrahydrofolate (CH3THF) for the remethylation
of homocysteine back to methionine. Other abbreviations: DMG, dimethylglycine;
Ado, adenosine; THF, tetrahydrofolate; CH2THF, 5,10-methylenetetrahydrofolate;
SO4, sulfate.
The determinants of
mild hyperhomocysteinemia, commonly seen in patients with cardiovascular
disease, are multifactorial and involve both genetic and acquired components.
Gene-nutrient interactions such as homozygosity for thermolabile MTHFR,
and low-folate nutritional status, can result in mild hyperhomocysteinemia.
Approximately 12 percent of the Caucasian population is homozygous for
thermolabile MTHFR. Smoking, excessive coffee consumption and lack of
exercise are associated with elevations in homocysteine as well.
What is hyperhomocysteinemia
and how is it determined? The term can be defined simply as "elevated
blood homocysteine" but the actual situation is more complex. When homocysteine
is transported out of cells into circulation,
it reacts with other compounds containing sulfhydryl (-SH) or disulfide
(-S-S-) groups. As a result of these reactions almost all of the homocysteine
in circulation is converted to a disulfide (oxidized) form. Less than
1 percent of total plasma homocysteine is found as the free -SH form.
The disulfide forms include the symmetrical dimer homocystine and mixed
disulfides with cysteine and cysteine-containing plasma proteins (Figure
3). In fact, over 70 percent of circulating homocysteine is carried as
a mixed disulfide by plasma proteins.
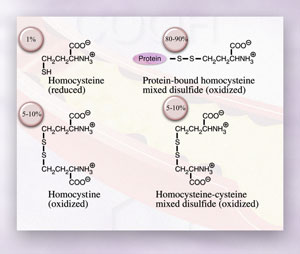
Figure 3. The circulating forms of homocysteine
that make up plasma total homocysteine.
Sensitive and reliable
assays for plasma total homocysteine (tHcy) were developed in the mid-
to late 1980s. This technical achievement was largely responsible for
establishing homocysteine as a major independent risk factor
for cardiovascular disease. In practice, plasma samples are treated with
strong reducing agents to break disulfide bonds, thus liberating free
homocysteine and other small thiols such as cysteine and glutathione.
The thiols are usually derivatized with a reporter group, separated and
detected. Thiol-specific fluorescent reporter groups are commonly used,
and separations are usually achieved by high- performance liquid chromatography
(HPLC), after which the compounds are detected fluorometrically (HPLC-FD).
Other methods use HPLC with electrochemical detection (HPLC-ED), or gas
chromatography with mass spectrometry (GC/MS). Immunoassays for plasma
tHcy were introduced about five years ago.
What is a "normal"
plasma tHcy? Until recently, the normal range for plasma tHcy was
considered to be 5 to 15 µmol/L. It is now widely accepted that the upper
limit of normal may be around 10 µmol/L for middle-aged adults and that
risk for cardiovascular disease occurs if plasma tHcy exceeds this value.
However, it is now also recognized that homocysteine levels increase with
age, perhaps as a result of micronutrient deficiencies due to malabsorption.
In the future, it is likely that age-specific reference ranges will be
established. Premenopausal women have approximately 20 percent lower values
than their male counterparts, suggesting that homocysteine metabolism
may be regulated to some extent by hormones. Patients with coronary artery
disease and other cardiovascular diseases usually have mild hyperhomocysteinemia
(>10 to 25 µmol/L) with an incidence of 30 to 50 percent. Almost all patients
with end-stage renal disease have hyperhomocysteinemia that tends to be
of an intermediate form (>25 to 50 µmol/L). Little or no homocysteine
is excreted by the normal kidney. The role of the kidney in homocysteine
metabolism and the regulation
of homocysteine metabolism is poorly understood. The homocystinurias,
rare inborn errors of homocysteine metabolism, are associated with severe
hyperhomocysteinemia (>50 to 500 µmol/L) and premature atherosclerosis
and thrombosis.
How does homocysteine
injure blood vessels? Because homocysteine is a thiol, it can undergo
autooxidation and oxidation with other thiols. The resulting reactive
oxygen species--hydrogen peroxide and superoxide anion radical—generate
oxidative stress. The concentration of plasma total cysteine is 20 to
30 times higher than that of plasma tHcy, yet cysteine, which also undergoes
similar oxidative reactions, is not usually considered a risk factor.
If oxidative stress is not the mechanism for homocysteine-induced vascular
dysfunction, is there perhaps another, more attractive hypothesis? Yes,
and it is related to direct molecular targeting by homocysteine. Recent
evidence suggests that homocysteine may limit the bioavailability of nitric
oxide, resulting in the impairment of flow-mediated vasodilatation. The
limited bioavailability of nitric oxide could be due to nitrosothiol formation
with homocysteine. Homocysteine may also target specific proteins and
impair their activity and function through disulfide bond formation. The
decreased binding of tissue plasminogen activator to homocysteine-modified
annexin II is a case in point and may explain, in part, the procoagulant
activity of homocysteine. Finally, as shown in Figure 4, homocysteine
may induce the expression and secretion of chemokines such as monocyte
chemoattractant protein 1 (MCP-1) and interleukin 8 (IL-8) in vascular
endothelial cells. Production of these chemokines by stimulated endothelial
cells would attract monocytes and neutrophils to sites of vascular injury
where they could take up residence in the intimal space.
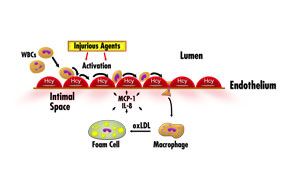
Figure 4. One proposed mechanism for
homocysteine (Hcy) involvement in vascular disease. White blood cells
(WBC) such as monocytes and neutrophils flowing through blood vessels
normally have random contact with vascular endothelial cells (EC). When
damage to ECs results from injurious agents, however, these WBCs begin
to roll along, and adhere to, the endothelial surface. Homocysteine may
speed the progression of vascular disease by stimulating production of
monocyte and neutrophil chemoattractants—MCP-1
and IL-8—in
the vascular endothelium. Secretion is targeted to the bottom side of
the cell, thereby establishing a concentration gradient for chemotaxis.
Once attached, monocytes migrate between ECs and become resident in the
vascular intimal space. Here they are transformed into macrophages, engulf
oxidized low-density lipoprotein (LDL), and become foam cells (the early
observed lesion called a fatty streak). Foam cells are a source of reactive
oxygen species which can play a role in other sequences of events that
promote atherosclerosis.
Is hyperhomocysteinemia
a treatable disease? Once a diagnosis of hyperhomocysteinemia has
been made, it is safe and easy to lower plasma tHcy in most individuals.
A cocktail of folic acid (400 to 800 µg), vitamin B12 (100 to 500 µg)
and vitamin B6 (25 to 100 mg) will reduce plasma tHcy up to
40 percent in subjects with cardiovascular disease. Whether lowering homocysteine
will have a beneficial effect on disease progression will be known in
3 to 5 years after the completion of a dozen or so worldwide clinical
trials involving over 70,000 subjects.
Should everyone
be tested for plasma tHcy? The American Heart Association has recommended
that individuals with a family history of heart and cardiovascular disease
be tested for plasma tHcy. Other subjects who should be tested are those
with premature atherosclerosis or atherosclerosis with no known conventional
risk factors such as hypertension or hyperlipidemia. Hypercoaguable profiles
now routinely include plasma tHcy. Of growing concern is the increased
incidence of cognitive dysfunction disorders, such as vascular dementia
and Alzheimer's disease, and the possibility that micronutrient deficiencies
resulting in hyperhomocysteinemia play a causative role. It may be common
practice in the near future to test everyone over the age of 60 for plasma
tHcy.
Additional Reading
Hajjar KA, Mauri L, Jacovina AT, Zhong FM, Mirza UA, Padovan JC, et al.
Tissue plasminogen activator binding to the annexin II tail domain. Direct
modulation by homocysteine. J Biol Chem 1998;273:9987-93.
Jacobsen DW. Homocysteine
and vitamins in cardiovascular disease. Clin Chem 1998;44(8 Pt 2):1833-43.
Jacobsen DW. Hyperhomocysteinemia
and oxidative stress: time for a reality check? Arterioscler Thromb Vasc
Biol 2000;20:1182-4.
Lentz SR. Mechanisms
of thrombosis in hyperhomocysteinemia. Curr Opin Hematol 1998;5(5):343-9.
Mansoor MA, Svardal
AM, Ueland PM. Determination of the in vivo redox status of cysteine,
cysteinylglycine, homocysteine, and glutathione in human plasma. Anal
Biochem 1992;200(2):218-29.
Refsum H, Ueland P,
Nygård O, Vollset SE. Homocysteine and cardiovascular disease. Annu Rev
Med 1998;49:31-62.
Robinson K, Mayer
EL, Miller DP, Green R, van Lente F, Gupta A, et al. Hyperhomocysteinemia
and low pyridoxal phosphate. Common and independent reversible risk factors
for coronary artery disease. Circulation 1995;92:2825-30.
Robinson K, Gupta
A, Dennis V, Arheart K, Chaudhary D, . . . Jacobsen DW. Hyperhomocysteinemia
confers an independent increased risk of atherosclerosis in end-stage
renal disease and is closely linked to plasma folate and pyridoxine concentrations.
Circulation 1996;94:2743-8.

|