DPC’s
Third Generation PSA assay gives clinicians the ability to use one PSA test
for all patient applications. It has assumed an indispensable role within
the realm of therapeutic PSA monitoring, prostate cancer therapy optimization
and outcome prediction due to its precision at very low serum concentrations.
In addition to providing accurate and timely quantification of PSA levels
during and after therapy, DPC’s Third Generation PSA assay is also well
suited for general prostate cancer screening purposes.
Prostate
cancer screening controversy
According
to the American Cancer Society (ACS), men aged 50 and older who receive
an annual medical examination should be offered a digital rectal examination
and a PSA test.1 The ACS also recommends that men aged 40 and
over be informed about the risk of prostate cancer2 and that
screening be considered at an earlier age, such as 40 or 45, in high-risk
individuals, e.g., African-Americans and first-degree relatives of prostate
cancer patients. In contrast, a number of physician organizations do not
advocate routine screening.3,4 This position is based on their
claims that there is no conclusive evidence that early detection and treatment
influence the overall death rate from this disease,5 and that
the standard treatment (radical prostatectomy) is associated with a high
incidence of side effects.6 Despite the conflicting recommendations,
PSA testing has increased rapidly among asymptomatic men in the US,7
and ACS statistics show a remarkable decline in the death rate from prostate
cancer over the past 5 years (Figure 1).8
Other
technologies may also be contributing to earlier detection of prostate
cancer. These include the availability of free PSA and use of the free-to-total
PSA ratio; improved ultrasonography and prostate biopsy methods; and the
wider application of risk assessment algorithms involving, for example,
age-adjusted PSA, prostate gland volume, transition zone volume and PSA
density. The decline in prostate cancer mortality may also be explained,
in part, by improved treatments for early-stage disease and better follow-up
care, as reviewed below.
Current
PSA studies indicate that cancer cases exhibit a slow linear PSA phase
initially, followed by a rapid exponential phase, and that PSA levels
begin to increase exponentially approximately 7 to 9 years before diagnosis.9
Experts agree on the benefits of calculating PSA doubling times (PSADT),
which are derived from a baseline measurement and subsequent PSA values
obtained at appropriate intervals. Published data indicate that the median
PSADT for localized prostate cancer is 4 years, but it is shorter in higher
clinical stages and worse histologic grades.10,11 In contrast,
PSADT in age-matched controls is much longer.12
Because
the IMMULITE® and IMMULITE® 2000 Third Generation
PSA assays possess outstanding low-end precision, they are ideal tools
for following PSA trends over time and obtaining accurate PSADT calculations.
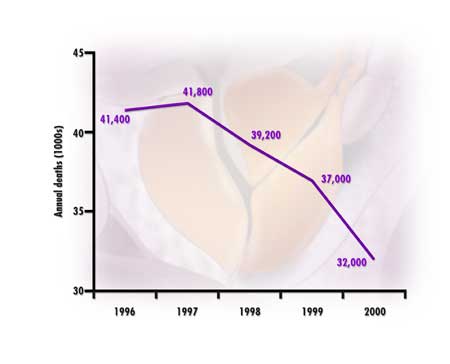
Figure
1. Five-year trends in prostate cancer. Based on collated 1995-2000
mortality statistics from the American Cancer Society. 8
Monitoring
treatment
The most generally useful clinical application of PSA determinations is
in the monitoring of treated prostate cancer patients. Treatments include
(radical) prostatectomy, radiation therapy, and hormonal therapies involving
medical or surgical castration. Circulating PSA levels can provide invaluable
information about the effectiveness of the therapy used and the existence
of residual cancer, as well as early detection of cancer recurrence. Rising
PSA levels can signal cancer activity well before any clinical signs of
recurrence appear. This lead time can be further increased by months or
even years when using ultrasensitive PSA immunoassays such as the IMMULITE
and IMMULITE 2000 Third Generation PSA assays. The expected PSA results
associated with good outcomes following various prostate cancer treatments
are listed in Table 1.
Studies
using DPC’s Third Generation PSA assays have established the value of
highly sensitive PSA assays to detect early prostate cancer relapse following
radical prostatectomy.13-15 Witherspoon et al. demonstrated
an average 18-month lead time in detecting disease progression using DPC’s
IMMULITE Third Generation PSA compared to a conventional PSA assay (Figure
2).13 Vassilikos et al. confirmed this lead time and was able,
over a 4-year follow-up period, to define a group of patients having rapid
PSA relapse.14 Doherty et al. showed that a single baseline
PSA level < 0.01 µg/L measured 4 to 6 weeks postsurgery indicates a lower
probability of disease progression.15 On multivariate analysis,
failing to achieve a PSA nadir of
< 0.01 µg/L was found to be an independent predictor of relapse, and a
stronger factor than even the Gleason sum or pathological margin status.

Figure
2. Postoperative PSA levels over time in a 73-year-old man who underwent
radical prostatectomy. No tumor was present at the surgical margins, seminal
vesicles or regional lymph nodes; and the postoperative baseline PSA was
0.004 µg/L. PSA at 4.2 years after prostatectomy became detectable at
0.10 µg/L using a conventional PSA assay. Using the IMMULITE Third Generation
PSA assay, a PSA rise was observed 2 years earlier.13
Definitive
radiation therapy in which the gland remains in situ rarely results in
an undetectable PSA, even when cure is achieved.19 However,
there is a general consensus that PSA levels reaching a nadir < 1.0 µg/L
are associated with improved biochemical disease-free survival.16,17
Disease progression is strongly suspected if the PSA rises on consecutive
determinations. The use of a highly sensitive PSA assay, such as the IMMULITE
or IMMULITE 2000 Third Generation PSA, can help detect early disease progression
in radiation-treated patients.
For
patients suffering PSA progression following primary surgery, viable treatment
options include salvage radiation or androgen deprivation therapy (ADT).
Salvage radiation treatment will benefit only those patients with proven
residual cancer in the prostatic fossa, whereas ADT can benefit those
with residual cancer and/or metastatic disease. Serial PSA measurements
in the months following surgery can be used to determine whether a patient
is more or less likely to benefit from salvage radiation therapy.18-20
Undetectable
baseline PSA levels defined (by conventional assays) as < 0.2 µg/L following
surgery, which later become detectable and progressively rise, suggest
locally recurring cancer in the prostatic fossa.18 (See Figure
2 for an example.) On the other hand, detectable PSA levels at baseline
which show progressive increases may represent microscopic metastatic
disease that was present prior to radical prostatectomy. In the former
scenario, salvage radiation treatment may be indicated, whereas systemic
salvage treatment using ADT would appear to be indicated in the latter.
Early PSA relapse, unfavorable Gleason sum and rapid PSADT are also associated
with prostate cancer metastases following both surgery and radiation treatment.26,27
The lead time afforded by ultrasensitive PSA assays and their superior
low-end precision identifies patients with early disease relapse and can
help guide medical decision-making regarding salvage therapy options.
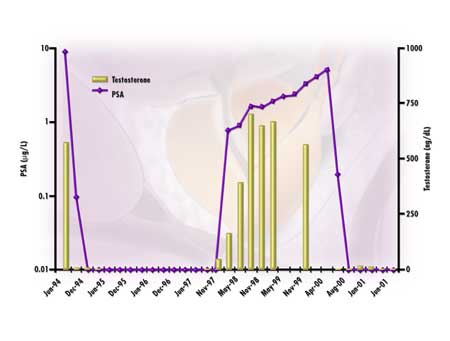
Figure
3. Time
course of PSA and testosterone levels in a 65-year-old patient with stage
T2a prostate cancer (Gleason sum 3 + 2 = 5 disease) treated with intermittent
androgen deprivation (IAD). Because therapy induced a rapid decline in
circulating PSA to persistently undetectable levels (<=0.01), androgen
deprivation was suspended in late 1997 and the subsequent rise in PSA
was monitored. When therapy was reinstated three years later, PSA levels
again dropped rapidly to undetectable levels. (Testosterone was measured
periodically to check the impact of therapy on circulating androgen levels.)
For
primary or salvage ADT, PSA is the most sensitive surrogate marker of
disease activity with which to judge treatment efficacy. Even in advanced
metastatic disease, achieving an undetectable PSA < 0.2 or < 0.3 µg/L
within 6 months of starting ADT, distinguishes patients with a favorable
and persisting response to ADT from those in whom only a limited response
can be expected.21,28 For patients with less extensive disease,
response to ADT is likely to be extremely favorable.22 Attainment
and maintenance of a PSA < 0.05 µg/L for a year or more while on ADT can
be used to identify patients with androgen-dependent prostate cancer,
in whom intermittent ADT is feasible.23-25
Patients
with low-volume disease, such as those having PSA relapse, can often enjoy
prolonged time-off treatment with improved quality of life.25 When retreatment
is indicated in such patients, due to a rising PSA or other clinical findings,
an excellent response to ADT can be anticipated.
Conclusion
It
has become essential for all-purpose PSA assays to have third-generation
capability. DPC’s Third Generation PSA assays effectively address the
requirements of modern prostate cancer treatment modalities by providing
extraordinary analytical sensitivity and a working range with excellent
precision at very low concentrations. At the same time, they accommodate
PSA values for general prostate cancer screening purposes.
Table
1. Relevant PSA values in various treatment modalities.

References
1. American Cancer Society. Man to man newsletter. The PSA blood test
and prostate cancer. Atlanta, GA: The Society, 2000.
2.
American Foundation for Urologic Disease. Prostate cancer resource guide.
Baltimore, MD: The Foundation, 1999. .
3.
US Preventative Services Task Force. Guide to clinical preventive services.
2nd ed. Washington, DC: US Dept. of HHS, PHS, Science, Office of Disease
Prevention and Health Promotion, Inc., 1996.
4.
American Cancer Society: Prostate cancer and cancer detection guidelines,
1999. Atlanta, GA: The Society, 1999.
5.
Centers for Disease Control and Prevention. Screening with the prostate-specific
antigen test-Texas 1997. Morbid Mortal Weekly Rep 2000 Sept 15;49(36).
6.
Stanford JL, Feng Z, Hamilton AS, Gilliland FD, Stephenson RA, Eley JW,
Albertsen PC, Harlan LC, Potosky AL. Urinary and sexual function after
radical prostatectomy for clinically localized prostate cancer: the Prostate
Cancer Outcomes Study. JAMA 2000 Jan 19;283(3):354-60.
7.
Potosky AL, Miller BA, Albertsen PC, Kramer BS. The role of increasing
detection in the rising incidence of prostate cancer. JAMA 1995 Feb 15;273(7):548-52.
8.
American Cancer Society. Mortality Statistics, 1995-2000. From the Web
site of the American Cancer Society: http://www.cancer.org
9.
Crawford ED, DeAntoni EP, Ross CA. The role of prostate-specific antigen
in the chemoprevention of prostate cancer. J Cell Biochem Suppl 1996;25:149-55.
10.
el-Galley RE, Petros JA, Sanders WH, Keane TE, Galloway NT, Cooner WH,
et al. Normal range prostate-specific antigen versus age-specific prostate-specific
antigen in screening prostate adenocarcinoma. Urology 1995 Aug;46(2):200-4.
11.
Schmid HP. Tumour markers in patients on deferred treatment: prostate
specific antigen doubling times. Cancer Surv 1995;23:157-67.
12.
Carter HB, Pearson JD, Metter EJ. Longitudinal evaluation of prostate-specific
antigen levels in men with and without prostate disease. JAMA 1992;267(16):2215-20.
13.
Witherspoon LR, Lapeyrolerie T. Sensitive prostate specific antigen measurements
identify men with long disease-free intervals and differentiate aggressive
from indolent cancer recurrences within 2 years after radical prostatectomy.
J Urol 1997 Apr;157(4):1322-8.
14.
Vassilikos EJ, Yu H, Trachtenberg J, Nam RK, Narod SA, Bromberg IL, Diamandis
EP. Relapse and cure rates of prostate cancer patients after radical prostatectomy
and 5 years of follow-up. Clin Biochem 2000 Mar;33(2):115-23.
15.
Doherty AP, Bower M, Smith GL, Miano R, Mannion EM, Mitchell H, et al.
Undetectable ultrasensitive PSA after radical prostatectomy for prostate
cancer predicts relapse-free survival. Br J Cancer 2000 Dec;83(11):1432-6.
16.
Lee WR, Hanlon AL, Hanks GE. Prostate specific antigen nadir following
external beam radiation therapy for clinically localized prostate cancer:
the relationship between nadir level and disease-free survival. J Urol
1996 Aug;156(2 Pt 1):450-3.
17.
Aref I, Eapen L, Agboola O, Cross P. The relationship between biochemical
failure and time to nadir in patients treated with external beam therapy
for T1-T3 prostate carcinoma. Radiother Oncol 1998 Aug;48(2):203-7.
18.
Cadeddu JA, Partin AW, DeWeese TL, Walsh PC. Long-term results of radiation
therapy for prostate cancer recurrence following radical prostatectomy.
J Urol 1998 Jan;159(1):173-7; discussion 177-8.
19.
Forman JD, Meetze K, Pontes E, Wood DP Jr, Shamsa F, Rana T, et al. Therapeutic
irradiation for patients with an elevated post-prostatectomy prostate
specific antigen level. J Urol 1997 Oct;158(4):1436-9; discussion 1439-40.
20.
Egawa S, Matsumoto K, Suyama K, Soh S, Kuwao S, Iwamura M. Limited suppression
of prostate-specific antigen after salvage radiotherapy for its isolated
elevation after radical prostatectomy. Urology 1999 Jan;53(1):148-54.
21.
Zagars GK, Sands ME, Pollack A, von Eschenbach AC. Early androgen ablation
for stage D1 (N1 to N3, M0) prostate cancer: prognostic variables and
outcome. J Urol 1994 May;151(5):1330-3.
22.
Fowler JE Jr, Pandey P, Seaver LE, Feliz TP, Braswell NT. Prostate specific
antigen regression and progression after androgen deprivation for localized
and metastatic prostate cancer. J Urol 1995 Jun;153(6):1860-5.
23.
Higano CS, Ellis W, Russell K, Lange PH. Intermittent androgen suppression
with leuprolide and flutamide for prostate cancer: a pilot study. Urology
1996 Nov;48(5):800-4.
24.
Grossfeld GD, Small EJ, Carroll PR. Intermittent androgen deprivation
for clinically localized prostate cancer: initial experience. Urology
1998 Jan;51(1):137-44.
25.
Strum SB, Scholz MC, McDermed JE. Intermittent androgen deprivation in
prostate cancer patients: factors predictive of prolonged time off therapy.
Oncologist 2000;5(1):45-52.
26.
Fowler JE Jr, Pandey P, Braswell NT, Seaver L. Prostate specific antigen
progression rates after radical prostatectomy or radiation therapy for
localized prostate cancer. Surgery 1994 Aug;116(2):302-5; discussion 305-6.
27.
Pound CR, Partin AW, Eisenberger MA, Chan DW, Pearson JD, Walsh PC. Natural
history of progression after PSA elevation following radical prostatectomy.
JAMA 1999 May 5;281(17):1591-7.
28.
Stamey TA, Kabalin JN, Ferrari M, Yang N. Prostate specific antigen in
the diagnosis and treatment of adenocarcinoma of the prostate. IV. Anti-androgen
treated patients. J Urol 1989 May;141(5):1088-90.
|