  |
The
first time a host is exposed to an antigen, the resulting primary immune
response is relatively weak and transient. After several days, during
which the antigen is taken up by phagocytes and presented to the antigen-specific
T and B lymphocytes, antibodies to the antigen begin to appear in the
serum. Initially, these antibodies are mainly of the IgM subclass. Gradually,
however, IgM is superceded by IgG. Increases in IgG titer occur after
a few more days. If no further antigen is administered, the concentrations
of the specific antibodies peak and eventually fall to undetectable levels.
When
the host is exposed to the antigen a second time, a markedly different
kinetic pattern is observed. The period between exposure and the appearance
of detectable antibody is much shorter. Whereas IgM is initially predominant
in the primary response, IgG levels exceed those of IgM overwhelmingly
throughout the entire secondary immune response. The IgG also shows a
much more rapid rise in titer, a longer plateau, and a slower decline
than it does during the primary immune response. In some cases, the IgM
response is seen only in the primary immune response, while in others,
IgM antibodies may be seen in a secondary response that resembles the
primary immune response. Nonetheless, the IgM titer is dwarfed by the
high IgG titer. Such characteristics are important to consider when basing
clinical diagnosis on infectious disease serology.
Using
an avidity assay in conjunction with IgG and IgM antibody response provides
a much clearer indicator of acute infection. The problem with relying
on only IgG and IgM antibody response is that IgM can persist for months
after primary infection and, as already mentioned, can reappear during
recurrent infections. In addition, IgG antibodies formed in the secondary
antibody response are of higher avidity (binding strength) than those
of the primary response. Initially, the antibodies formed are mostly of
relatively low avidity. Over time, however, the antibodies with high avidity
for the antigen increase, while those with low avidity decrease in titer.
Can
the clinical laboratory exploit this phenomenon as an aid in diagnosis?
In the late 1980s, Hedman et al.1 introduced a new method of
detecting recently acquired Toxoplasma infection, based on the
strength of the binding of specific IgG to multivalent Toxoplasma
antigen. Toxoplasma gondii can cause serious damage to the
fetus when a pregnant woman acquires a primary infection. The presence
of Toxoplasma-specific IgM in serum as an indicator of recent infection
is unreliable, due to differences in the persistence of these antibodies.
In some patients, specific IgM may persist for months or even years following
a primary infection. Jenum et al.2 observed that women with a possible
acute infection have a low mean IgG avidity, based on a combination of
positive specific IgM and a high Sabin-Felman dye titer of greater than
300 IU/mL (the gold standard test). In women positive for Toxoplasma-specific
IgM, but with a low titer, the mean IgG avidity was high. In a third group
of women with latent infection without detectable Toxoplasma-specific
IgM, the mean IgG avidity was high also. The introduction of Toxoplasma-specific
IgG avidity testing on IgM-positive samples collected in the first trimester
of pregnancy can eliminate false-positive results that would suggest recent
infection. Thus, IgG avidity testing can prevent unnecessary anxiety,
as well as additional examinations and treatment.
In
my opinion, one can use this avidity assay in two ways: to improve the
diagnostic routine and to discriminate between primary and latent infection.
If IgG and IgM results are positive, one can perform an avidity assay
and, in the case of high avidity, rule out recent infection. A finding
of low avidity indicates the need for further testing by a reference laboratory.
Furthermore, all IgG-positive results should be followed by an avidity
assay. A high-avidity result rules out a recent infection, while a low-avidity
result should be followed by an IgM assay. In the case of a positive IgM
result, a recent infection is almost certain. A second sample should be
requested to establish a baseline, or the serum sample should be sent
to a reference laboratory for confirmation of results. (See Figure 1.)
Clinical laboratory diagnostics can now discriminate between primary and
latent infection more accurately by examining the different binding capacities
of IgG molecules.
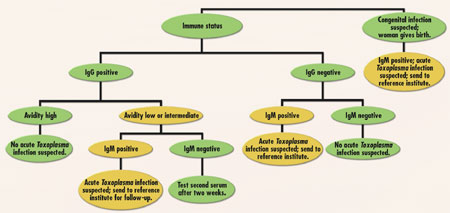
Figure
1. Toxoplasmosis testing algorithm in use at the Medical Center Alkmaar,
The Netherlands. (Reproduced with permission of F. Vlaspolder.)
|