  |
DPC
is pleased to be the first immunodiagnostics manufacturer to offer random
access automation of five key cardiac assays.
First available fully automated, random access
immunoassay for
|
|
Homocysteine
(IMMULITE 2000) = 0.97 (HPLC) + 0.71 µmol/L
Means
13.4 µmol/L
(IMMULITE 2000) r =
0.974
13.2 µmol/L
(HPLC) N
= 95 (Plasma Samples) |
homocysteine
DPC's IMMULITE 2000 Homocysteine assay quantitatively measures L-homocysteine
in serum or plasma, and can assist in the diagnosis and treatment of patients
suspected of having hyperhomocysteinemia or homocystinuria. It is easy
to perform and includes on-board sample pretreatment, eliminating the
need for a manual pretreatment step. IMMULITE 2000 Homocysteine showed
good correlation with an in-house HPLC method in use at a US reference
lab. (See figure.)
Wide-range, high-sensitivity CRP assays
|
|
|
High Sensitivity
CRP
(IMMULITE 2000) = 1.01 (Nephelometry) - 0.01 mg/dL
Means
0.23 mg/dL (IMMULITE 2000) r
= 0.994
0.24 mg/dL (Nephelometry) N
= 199 (Patient Samples)
|
DPC's IMMULITE and IMMULITE 2000 High Sensitivity CRP assays are intended
for the quantitative measurement of C-reactive protein in serum or plasma,
as an aid in the detection and evaluation of infection, tissue injury,
inflammatory disorders and associated diseases. Additionally, measurement
of CRP by high-sensitivity CRP assays may add to the predictive value
of other markers used to assess the risk of cardiovascular and peripheral
vascular diseases. The DPC assays have a functional sensitivity of 0.02
mg/dL (0.2 mg/L) and a calibration range of up to 25 mg/dL (250 mg/L).
They offer better sensitivity and a wider dynamic range than do conventional
CRP assays, while minimizing the cost and labor of additional sample dilutions
or calibrators required by other methods. The IMMULITE 2000 assay showed
close agreement with a commercial, high-sensitivity nephelometric method.
(See figure.)
Reliable and accurate troponin I assays
DPC offers assays for troponin I on three platforms. The IMMULITE and
IMMULITE 2000 systems, with their broad menus, allow for workstation consolidation;
and the IMMULITE Turbo assay provides STAT testing, delivering the first
result in 15 minutes. The assays measure troponin I in serum or in heparinized
or EDTA plasma, as an aid in the diagnosis of acute myocardial infarction
(AMI). Consistency is assured by the use of antibodies specific for the
stable portion of the troponin I molecule, and by equimolar measurement
of all troponin I forms. The US National Academy of Clinical Biochemistry
(NACB) has specified cardiac troponin as the best marker for definitive
AMI diagnosis.
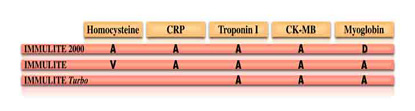
A = available worldwide
V = in validation
D = under development
Fast, faster and fastest CK-MB assays
DPC offers assays for CK-MB on all three IMMULITE platforms for the quantitative
measurement of creatine kinase isoenzyme MB (CK-MB) in heparinized plasma
or serum, as an aid in patient management and the assessment of prognosis
of myocardial infarction. The IMMULITE and IMMULITE 2000 CK-MB assays
require a single 30-minute incubation and have calibration ranges of up
to 500 ng/mL. The IMMULITE Turbo assay provides the first result in 15
minutes.
Myoglobin—a
rapid triage marker
DPC's IMMULITE Myoglobin assay is intended for the quantitative measurement
of myoglobin in serum or in heparinized or EDTA plasma, as an aid in the
diagnosis of acute myocardial infarction. When run in the Turbo mode,
it has a calibration range of up to 800 ng/mL, and delivers the first
result in 15 minutes. The NACB has specified myoglobin as the best early
marker for AMI and a highly effective tool for AMI rule-out.
Cardiac solutions
The IMMULITE family of immunoassay systems provides state-of-the-art support
for cardiac testing. Recognizing the trend toward workstation consolidation
and the importance of rapid turnaround, DPC offers cardiac solutions to
meet today's testing needs.

|